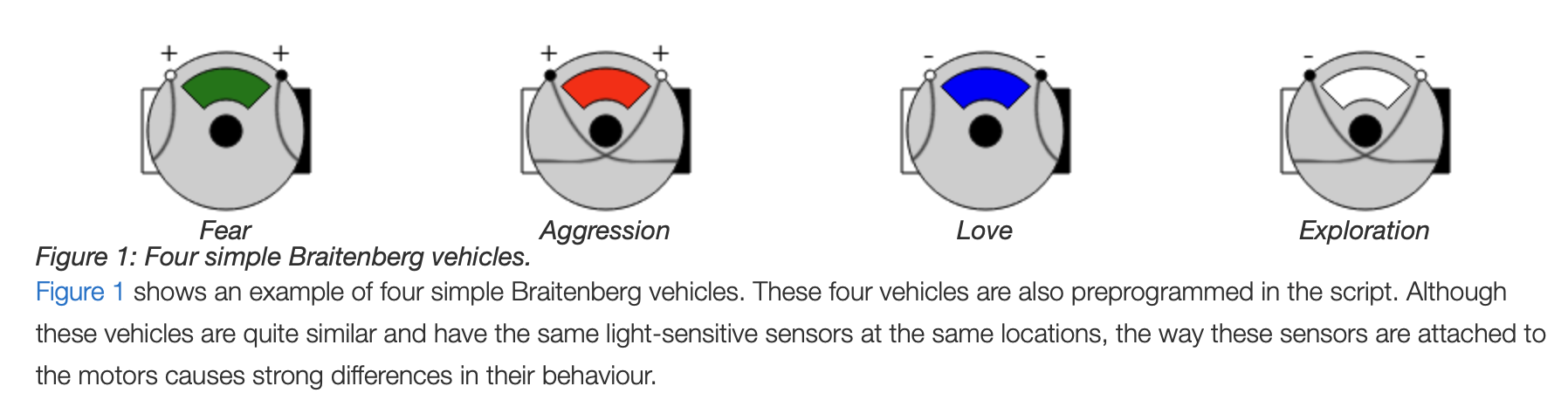
flowchart TD N[Nervous System] --> B[Body] B --> W[World]
Action
PSY 511.001 Spr 2026
Department of Psychology
Overview
Prelude
– Reviews (2012)
Announcements
- Exercise ANAT due today.
Today’s topics
- Warm-up
- On action
- How muscles work
- Controlling muscles
- Illustrative motor systems
Warm-up
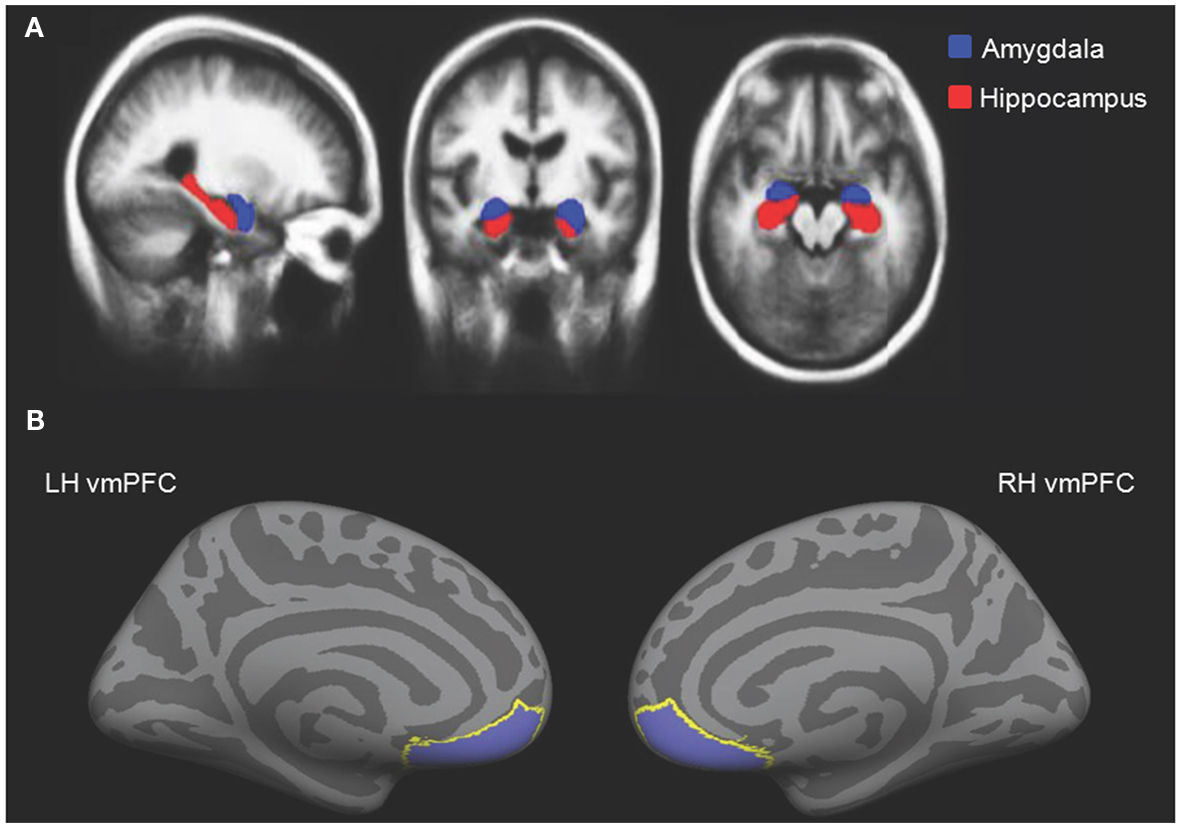
The amygdala is found deep within which lobe of the cerebral cortex?
- A. The occipital lobe
- B. The frontal lobe
- C. The insula
- D. The temporal lobe
The amygdala is found deep within which lobe of the cerebral cortex?
A. The occipital lobeB. The frontal lobeC. The insula- D. The temporal lobe
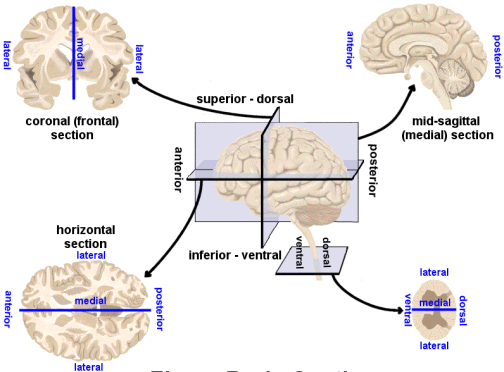
The middle image in A in the figure at right depicts what plane of section?
- A. Sagittal
- B. Dorsal
- C. Coronal
- D. Horizontal
The middle image in A in the figure at right depicts what plane of section?
A. SagittalB. Dorsal- C. Coronal
D. Horizontal
Which division of the peripheral nervous system (PNS) ennervates the gastrointestinal tract?
- A. Enteric
- B. Cerebellum
- C. Sympathetic
- D. Parasympathetic
Which division of the peripheral nervous system (PNS) ennervates the gastrointestinal tract?
- A. Enteric
B. CerebellumC. SympatheticD. Parasympathetic
On action
Why action?
“Quote origin” (n.d.)
“Quote origin” (n.d.)
Animals are animate
- Actions influence
- Their own bodies, \(B\)
- The external world, \(W\)
- Their own bodies, \(B\)
- The nervous system controls the body and its effects on the world
Laws govern movement
- Physics
- Biology
- Psychology
Your turn
Can you name a physical law that applies to movement? A biological law? A psychological law?
Suggestion: Fitts’ (1954) Law: Speed/accuracy trade-off
Movement one of nervous system outputs
- Somatic
- Autonomic
- Endocrine
flowchart TD N[Nervous System] -- autonomic --> B[Body]; N -- somatic --> B; N -- endocrine --> B;
Inputs
- Exteroceptive (outside the body)
- Interoceptive
flowchart TD B[Body] -->|interoception| N[Nervous System] B -->|exteroception| N
flowchart TD N[Nervous System] -- autonomic --> B[Body] N -- somatic --> B N --->|endocrine| B B --->|endocrine| N B --->|action| W[World] W --->|perception| B B --->|somatic| N B --->|autonomic| N
Computational perspective
Neuromechanics perspective
Evolutionary perspective
Evolutionary perspective
- Live long enough…
- Avoid injury & predation
- Secure sustenance
- Maintain health
- Reproduce…
- Find a mate
- Mate
- Ensure the well-being of offspring
Classes of behavior
- Orienting, info gathering
- Maintaining balance
- Reaching, grasping, & manipulation
- Locomotion
- Communication
- Mating & caregiving
- Ingestion & elimination
- Defense
- Sleep
Your turn
What’s missing?
The real reason for brains
– Wolpert (2011)
But simple circuits…
Even single cells…
– Totally Science (2021)
Engines of animal motion
How muscles work
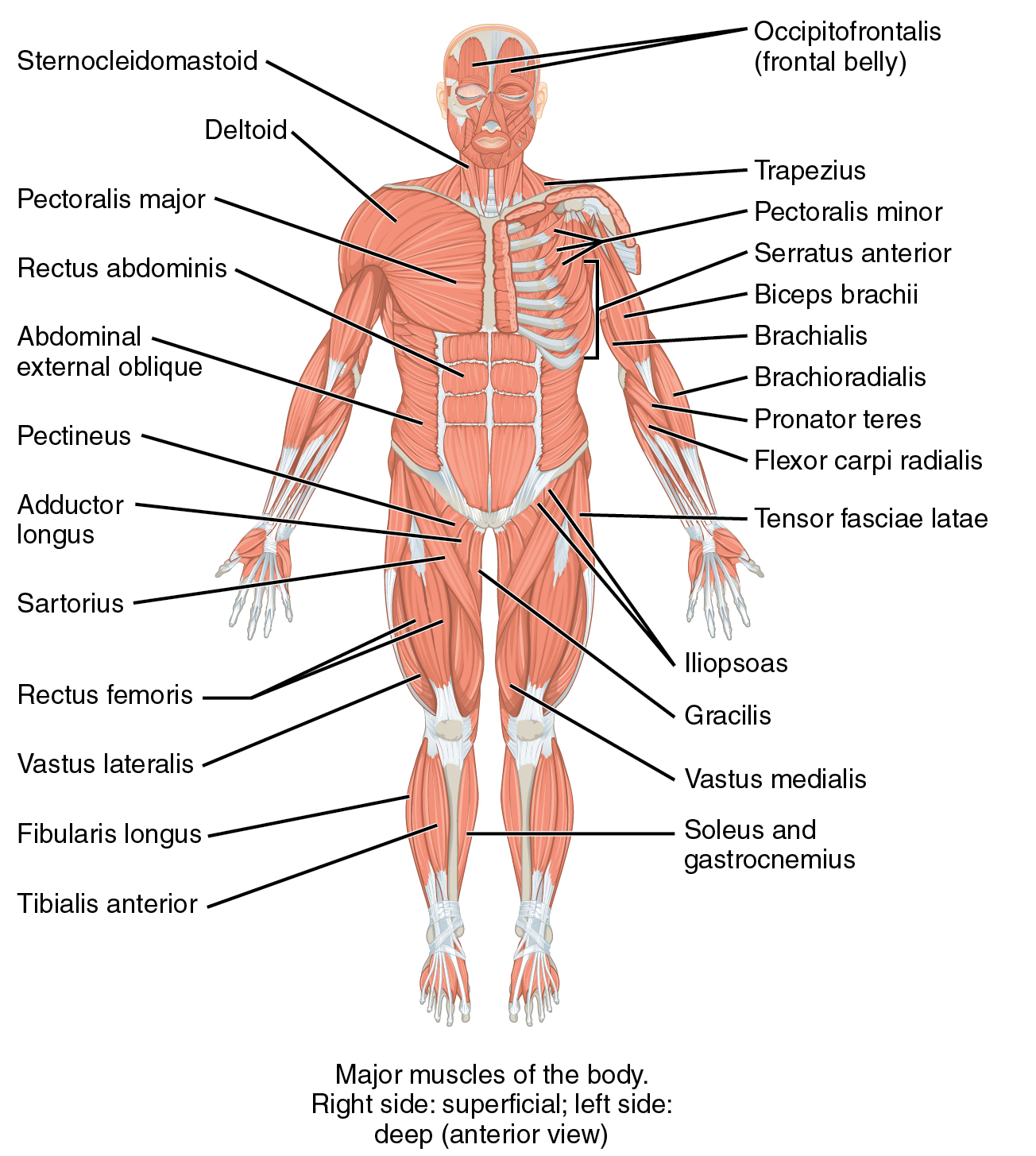
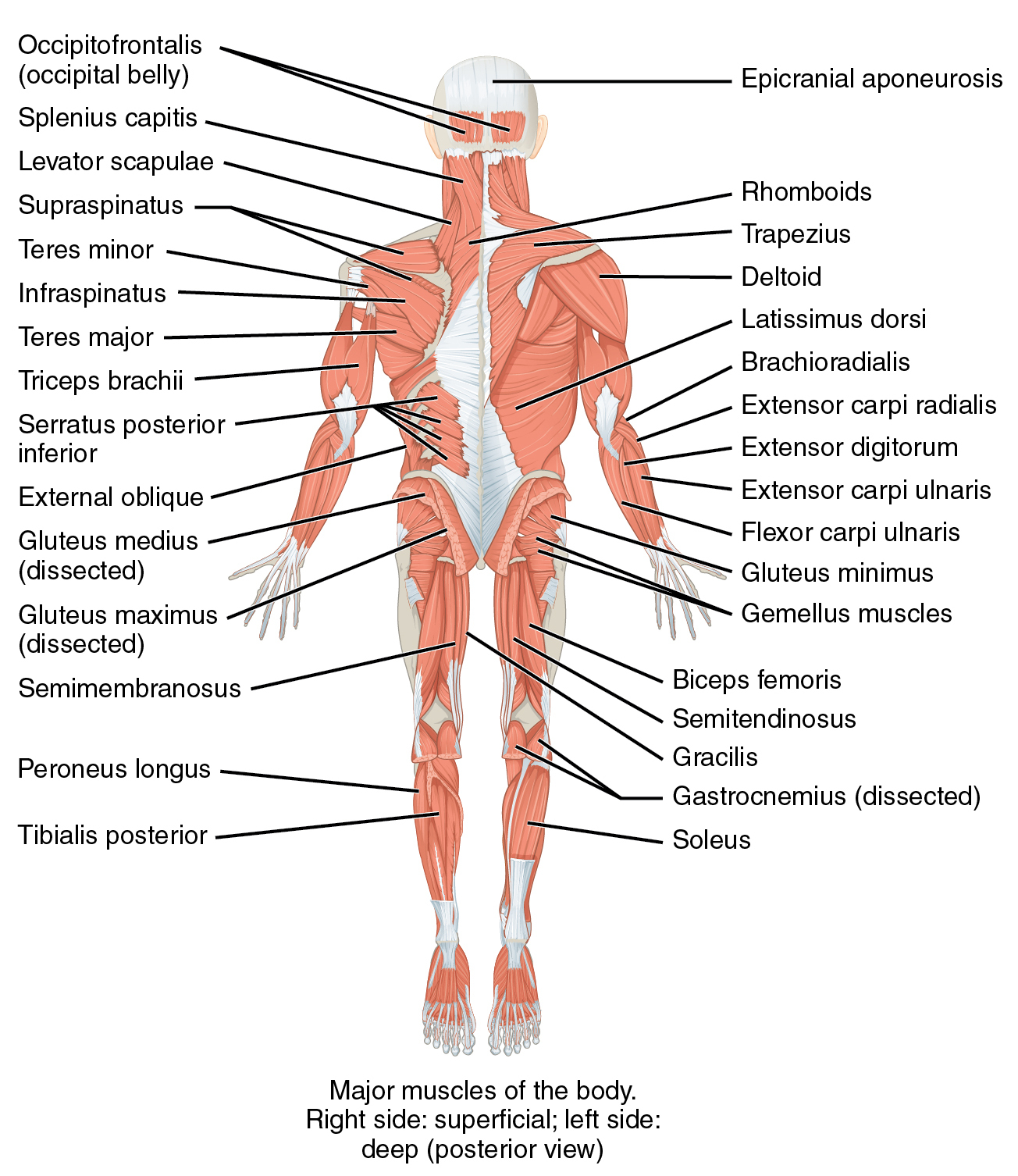
Muscles are machines that
Functional classes
- Axial
- Trunk, neck, hips
- Proximal
- Shoulder/elbow, pelvis/knee
- Distal
- Hands/fingers, feet/toes
Agonist/antagonist pairs
CNS must…
Generate signals that move axial, proximal, & distal muscles in coordination.
Generate signals that move agonist/antagonist muscle pairs in sequence.
How skeletal muscles contract
How skeletal muscles contract
Generating force
- More force
- \(\rightarrow\) faster stimulation rate
- Recruit (activate) more motor units
Fiber types
- Fast twitch/fatiguing
- Type II4
- White meat
- Slow twitch/fatiguing
- Type I
- Red meat
Muscles are sensory organs, too!
Stretch receptors
- Sense muscle length \(l\) and \(\Delta l\), e.g. “stretch”
- Muscle proprioception
- Proprioception == perception about the self
- A form of interoception
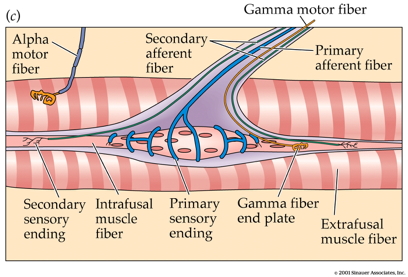
Intrafusal fiber connections
- Type Ia afferents5
- Info about length \(l\) and \(\Delta l\)
- Via stretch receptors (aka muscle spindles)
- Type II afferents
- Info about length \(l\) only
- Force-producing motor fiber (efferent)
- Motor input
- Ennervated by gamma (\(\gamma\)) motor neurons
Extrafusal fibers
- Generate force
- Ennervated by alpha (\(\alpha\)) motor neurons
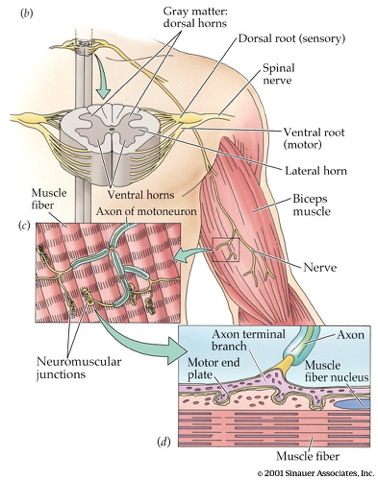
Skeletal muscle circuit
flowchart TD
N{Nervous System} ==>|Alpha motor neurons|B(Extrafusal muscle fibers)
N ==>|Gamma motor neurons|C(Intrafusal muscle fibers)
C ==>|Type Ia afferents|N
C ==>|Type II afferents|N
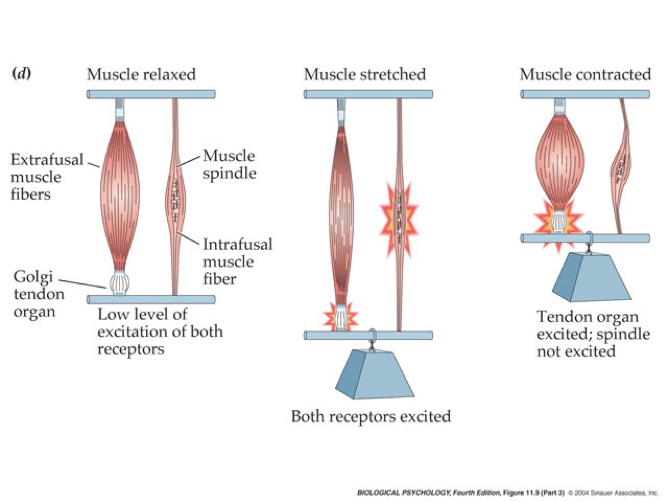
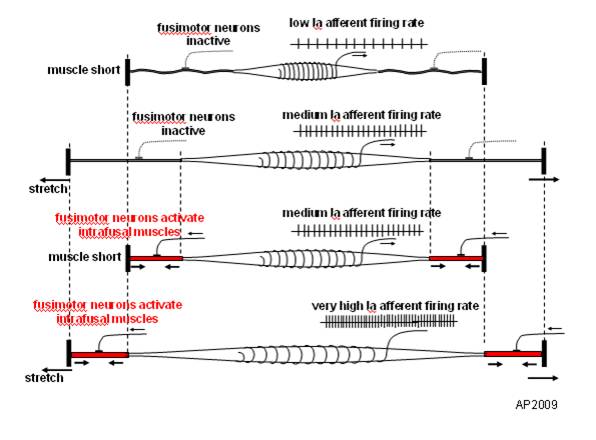
Monosynaptic myotatic stretch reflex
- Muscle stretched (length increases)
- Muscle spindle in intrafusal fiber activates
- Ia fiber \(\rightarrow\) spinal cord
- Activates alpha (\(\alpha\)) motor neuron
- 1 synapse \(\rightarrow\) monosynaptic
- \(\alpha\) motor neuron activates extrafusal fiber
- Muscle contracts, shortens length
- Gamma (\(\gamma\)) motor neuron fires to take up ‘slack’ in intrafusal fiber
Suppressing tremor
Why it matters…
Also: Patellar reflex
– 牛津基礎生物學 (2021)
Conduction speed
Interim summary
- Voluntary movements involve motor units causing contraction of agonist or antagonist pairs of skeletal muscles
- Skeletal muscles have embedded sensors that detect length/tension
- CNS gets rapid6 feedback about muscle length/tension (proprioception)
- Monosynaptic stretch reflexes maintain stability of limb/body position
Controlling muscles
Key nodes
- Primary motor cortex (M1)
- Anterior to central sulcus
- Frontal lobe
Somatotopic8 organization
Key nodes
- Non-primary motor cortex
- Supplementary motor area (SMA)
- Premotor cortex (PMC)
- Frontal eye fields (FEF)
Key nodes
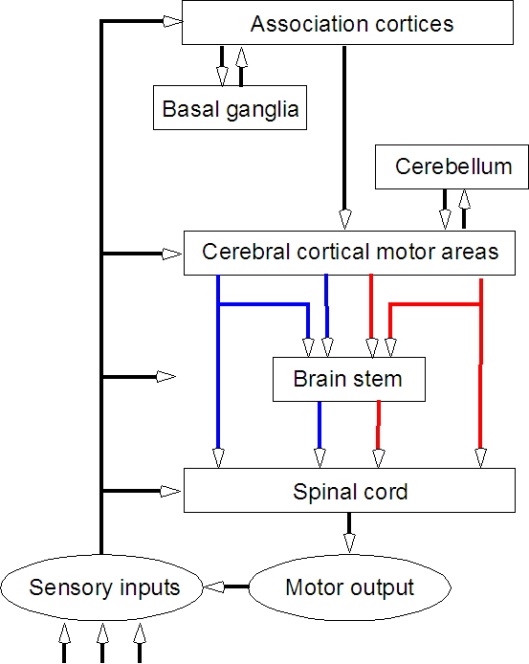
- Basal ganglia
- Brain stem
- Cerebellum
- Spinal cord
Projection pathways
- Pyramidal tracts/system
- Extrapyramidal system
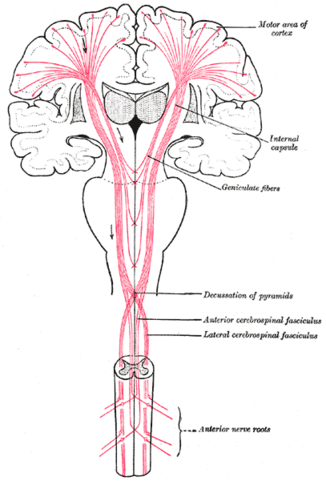
Pyramidal tracts/system
Pyramidal tracts/system
- Pyramidal cells
- Layer 5 in primary motor cortex (M1)
- Corticobulbar (cortex -> brainstem) tract
- Corticospinal (cortex -> spinal cord) tract
Pyramidal tracts/system
Spinal cord projections
- Separate ascending (afferent) and descending (efferent) pathways
- Ascending (generally) more dorsal; descending more ventral
- White matter on exterior (unlike cerebral cortex)
Somatotopic map & spinal cord segments
Dermatomes & myotomes
Direct cortical control
- Over some motor neurons
- In humans; prevalence uncertain in other animals
- For individuated (“fractionated”) movements of fingers, toes, lips, but other muscles, too.
Extrapyramidal system
- Involuntary movements
- Posture, balance, arousal
- Tectospinal tract
- Lateral Vestibulospinal tract
- Reticulospinal tract
- Rubrospinal tract
Neuromechanical17 perspective
- “Closed loop” control
- Physical intelligence (pre-flexes)
- Conduction speed slow
- Physical/mechanical (e.g., spring-like) properties of muscles, tendons
Illustrative motor systems
Behavioral repertoires
- Looking
- Locomotion
- Reaching & grasping
- Speaking
Approach
Looking
- Behavioral goals
- Direct gaze (eyes + head + body) toward goal-relevant targets
- Direct gaze toward emergent targets
- Track/pursue targets across observer/target motion
- Maximize quality of visual information about targets
Eye movement types
- Voluntary
- Saccades
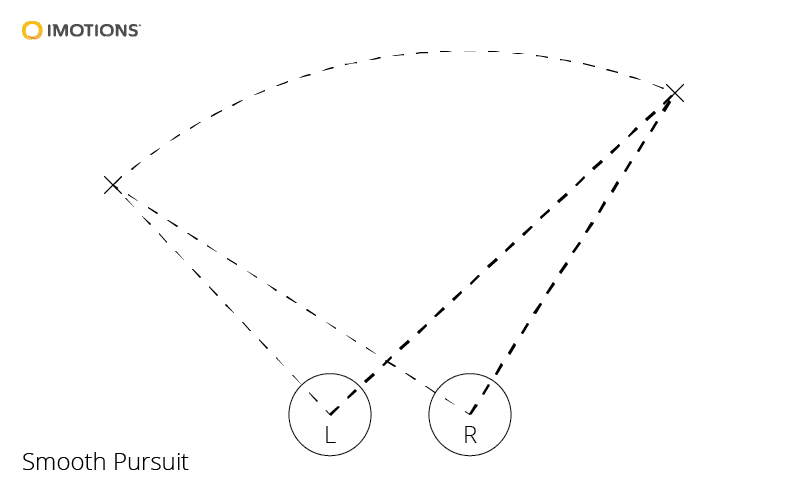
- Smooth pursuit
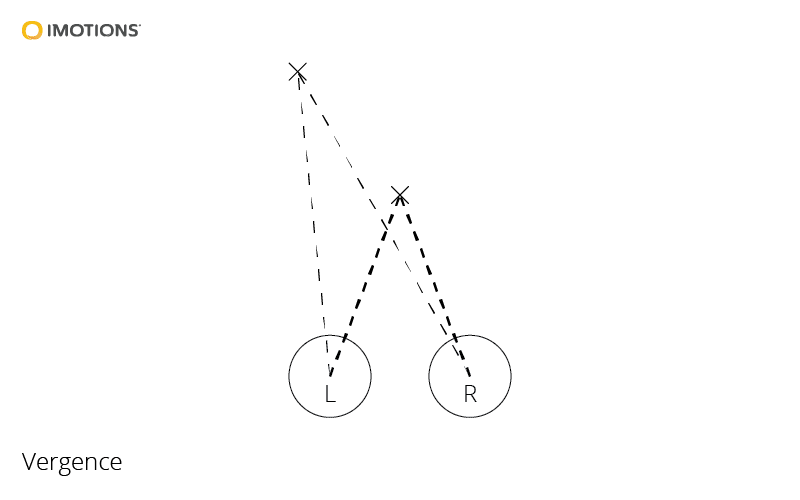
- Vergence
- Involuntary
- Vestibulo-occular reflex (VOR)
- Optokinetic nystagmus
- Accommodation
Saccades
- Rapid, ballistic (no online feedback)
- ~2-3/s
- ~200 ms to initiate
- Amplitudes >20 deg \(\rightarrow\) saccade + head movement
– Wikipedia contributors (2025b)
Smooth pursuit
Vergence
- Eyes move disjointly
- Fixate on comparable locations
- Binocular vision \(\rightarrow\) 3D depth
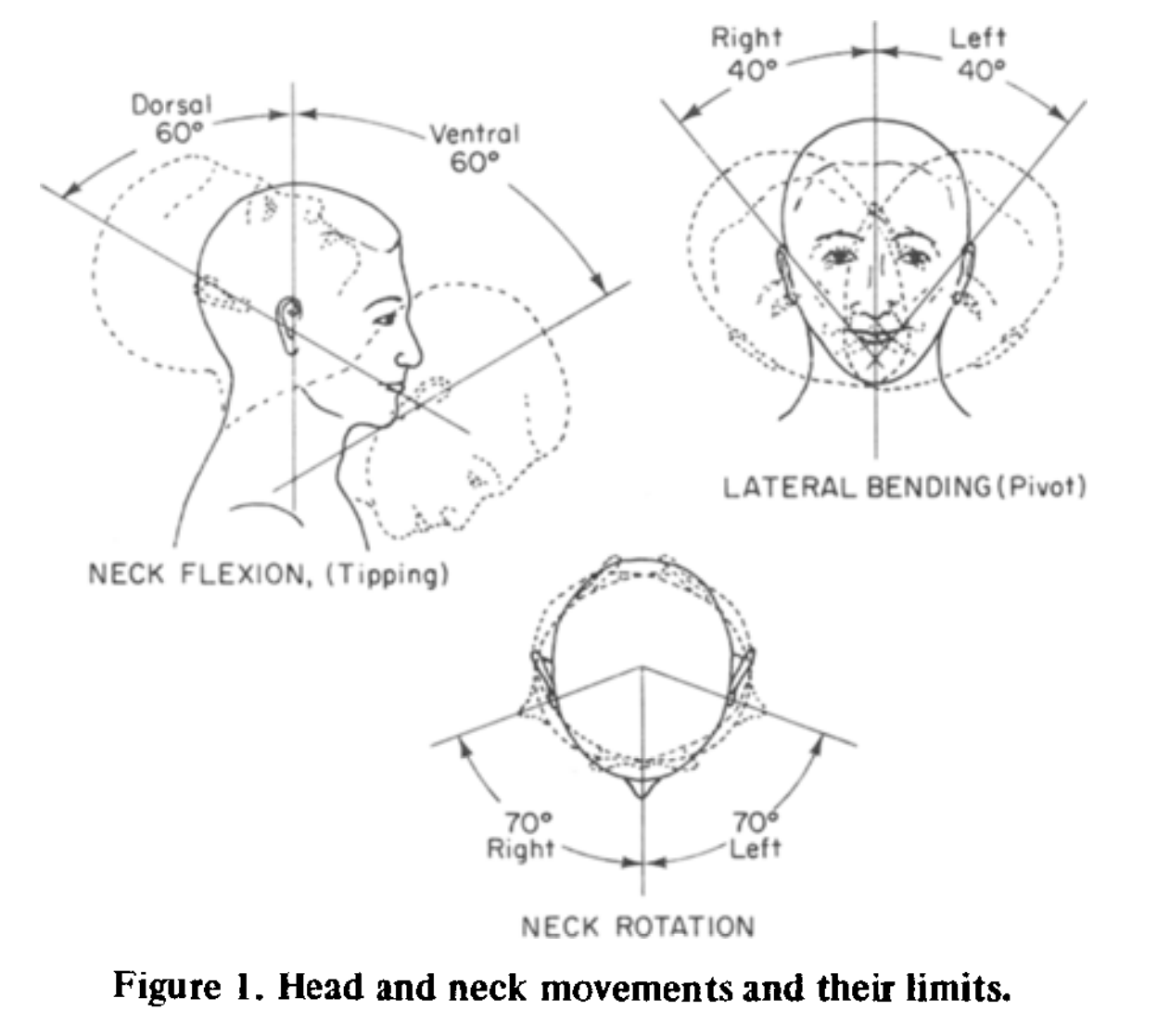
Head movements
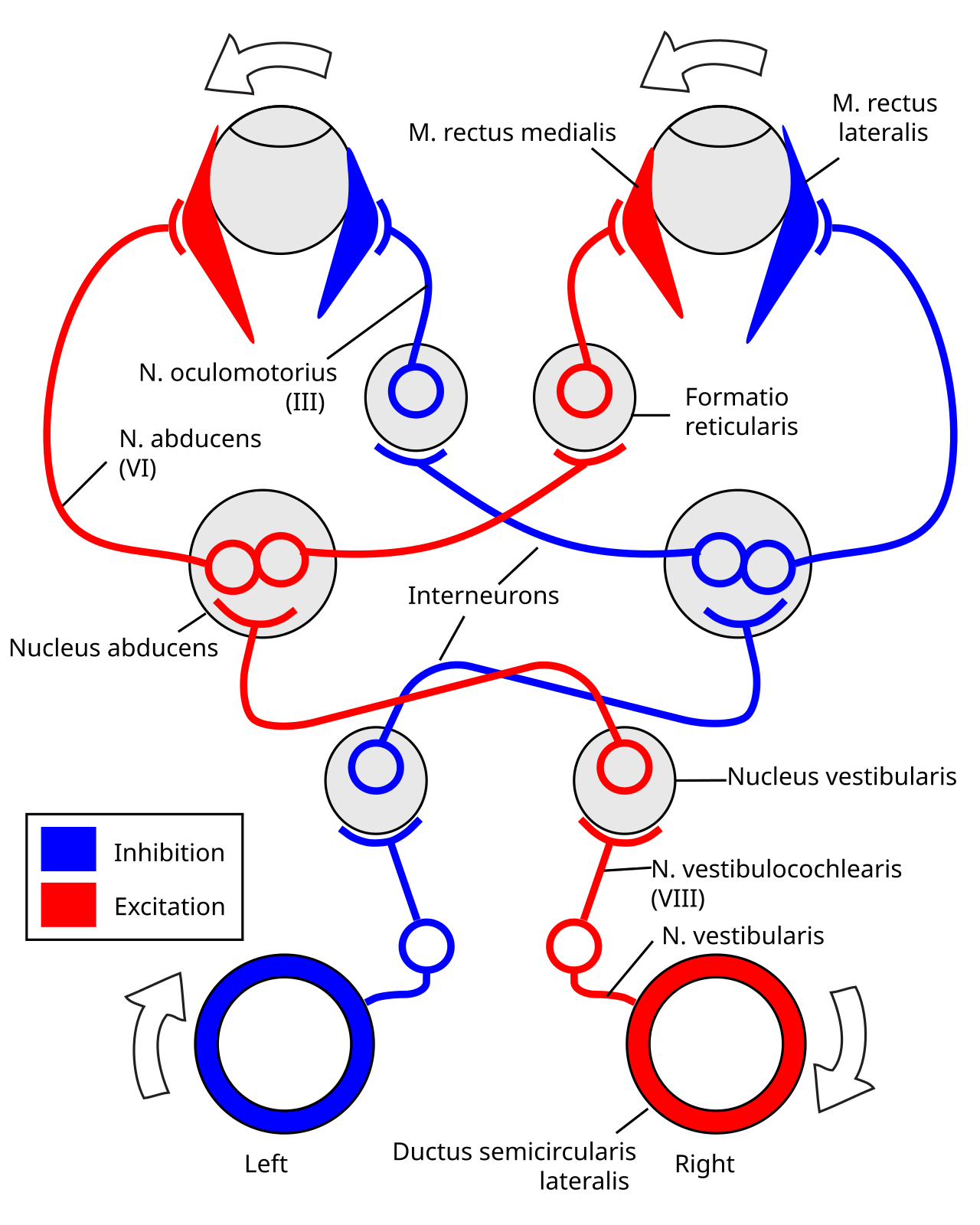
Vestibulo-occular reflex (VOR)
- Steady eyes when head moves
- Fast: response < 10 ms
- Disturbed in vertigo
Optokinetic nystagmus (OKN)
- Also called optokinetic response
- Involuntary fast tracking of whole field visual motion, then reset
Pupil constriction/dilation
- Bright light \(\rightarrow\) constriction
- Also by parasympathetic activation
- Dim light \(\rightarrow\) dilation
- Also by sympathetic activation
Eye muscles
Eye muscles
| Nerve | Eye Muscle |
|---|---|
| CN24 III Oculomotor | Superior rectus |
| Inferior rectus | |
| Medial rectus | |
| Inferior oblique | |
| Iris (pupillary) sphincter | |
| CN IV Trochlear | Superior oblique |
| CN VI Abducens | Lateral rectus |
| ANS sympathetic | Iris (pupillary) dilator |
Inputs to CN III
flowchart TD
A["Frontal Eye Fields"] -->|"Voluntary saccades"| B(("CN III"))
C["Superior Colliculus"] -->|"Reflexive saccades"| B
D["Vestibular (CN VIII) n."] -->|"VOR"|B
B --> E["Superior rectus"]
B --> F["Inferior rectus"]
B --> G["Medial rectus"]
B --> H["Inferior oblique"]
I["Parasympathetic NS"] -.-> B
B -.-> J["Iris (pupillary) sphincter"]
Considerations
- (Multi)sensory feedback
- Move eyes \(\rightarrow\) shift visual field
- Change vergence or lens thickness \(\rightarrow\) change image focus
- Eye muscles (probably) have proprioceptors analogous to spindles in skeletal muscles
Considerations
- Relatively simple physics:
- Limited influence of gravity, friction
- Small # of muscles \(\rightarrow\) “model” motor system?
- DEMO: Illustration of “forward”/generative model/unconscious inference?
- Predicting future/interpreting current sensory states
- Converging inputs: voluntary and involuntary control
Locomotion: Goals
- Keep balance
- Control speed
- Steer
- Toward
- Away
Physics of balance
- Body ~ inverted pendulum
- Inherently unstable
- Requires control
- Keep Center of Mass (COM) within base of support
Locomotion: Neuromechanics
Locomotion: Physics
Locomotion: Physics
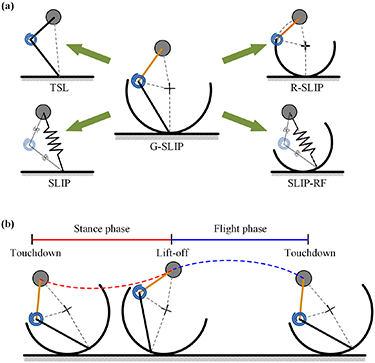
- Legs, hips can be modeled as generalized spring-loaded inverted pendulum (G-SLIP)
Locomotion: On steering
- Taxis
- Directed movement
- Toward (positive taxis)
- Away (negative taxis)
- Directed movement
- vs. Kinesis (random movement)
- Steering a key “breakthrough” in the history of intelligence (Bennett, 2023)
Locomotion: Neural signalling
- Animals with brain input to the spinal cord can locomote on a treadmill
- Central pattern generators (CPG) in spinal cord?
Or “rotational” patterns…
Speech: Goals
- Involuntary vs. voluntary
- Vocalizations types
- Linguistic
- Paralinguistic
- Pitch, tone, rhythm, tempo, etc.
- Non-linguistic
Components
- Lips, tongue, mouth
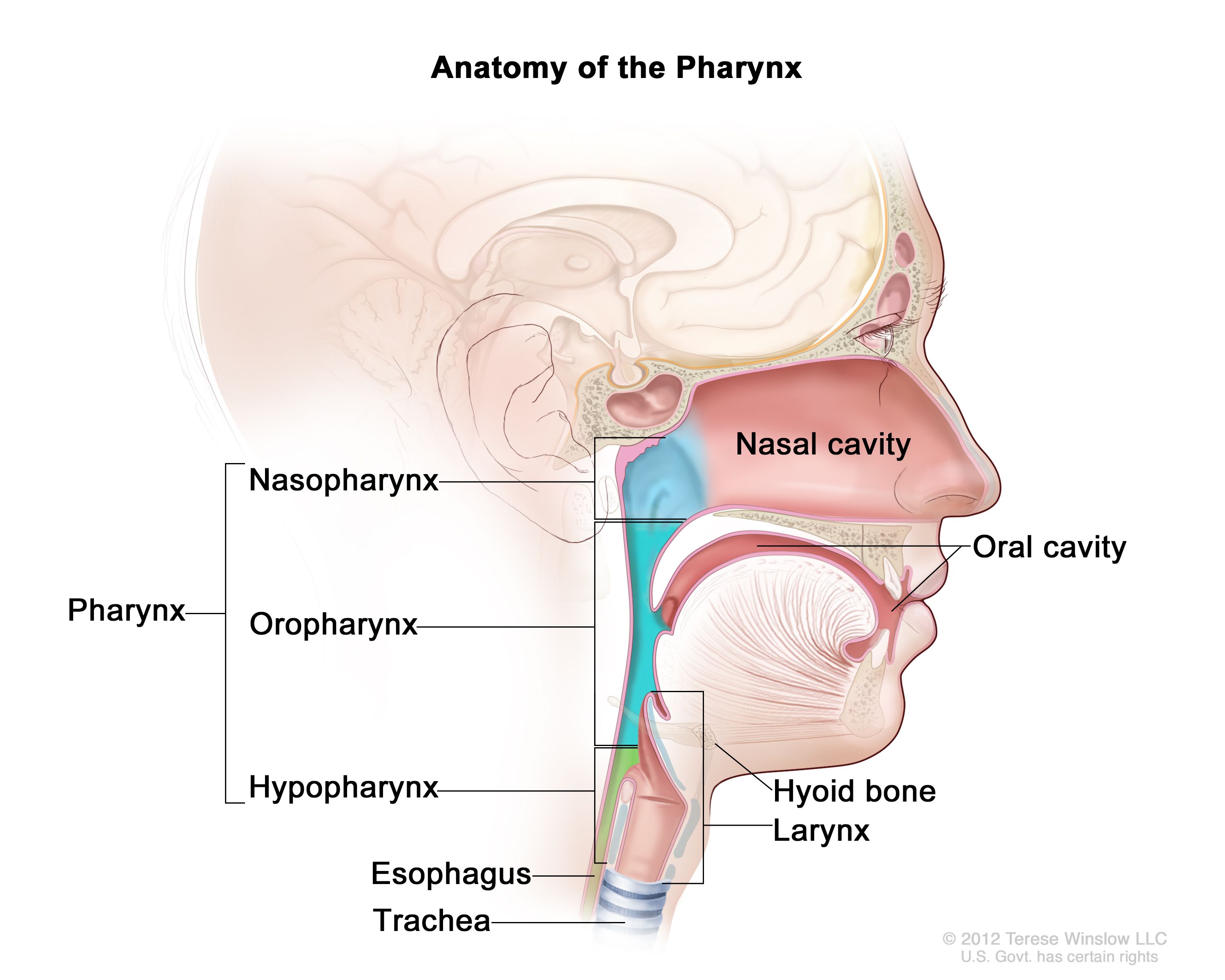
- Pharynx
- Larynx
- Lungs
https://en.wikipedia.org/wiki/File:Real-time_MRI_-_Speaking_(English).ogv
– Wikipedia contributors (2026)
Consonants
- Place and manner of articulation
- Voicing
- “B” vs. “P”
- “D” vs. “T”
- “G” vs. “K”
- “Z” vs. “S”
flowchart TD Z["Motor cortex"] & Y["Periaqueductal gray"] --> A & C & E & G A["CN XII Hypoglossal"] --> B["Tongue"] C["CN X Vagus"] --> D["Larynx"] E["CN VII Facial"] --> F["Lips & face"] G["CN IX Glossopharygeal"] --> H["Pharynx"]
Example
– VideoCollectables (2014)
Considerations
- Hierarchical, parallel control
- Sensory feedback
- Vocal muscle stretch receptors
- Auditory
- Vocal muscles like fingers
- Large area of M1
- Individuated control over
Illustrative motor systems: Interim summary
- Many (most?) behaviors involve sequences of
- looking/orienting + locomotion + reaching/manipulation + vocalization
- Parallel, coordinated, sequential outputs to multiple motor systems
Interim summary
- Direct (cerebral) cortical control over muscles of eyes, fingers, vocal apparatus
- Feedback critical, often multisensory
- Exploiting physics of body
Wrap-up
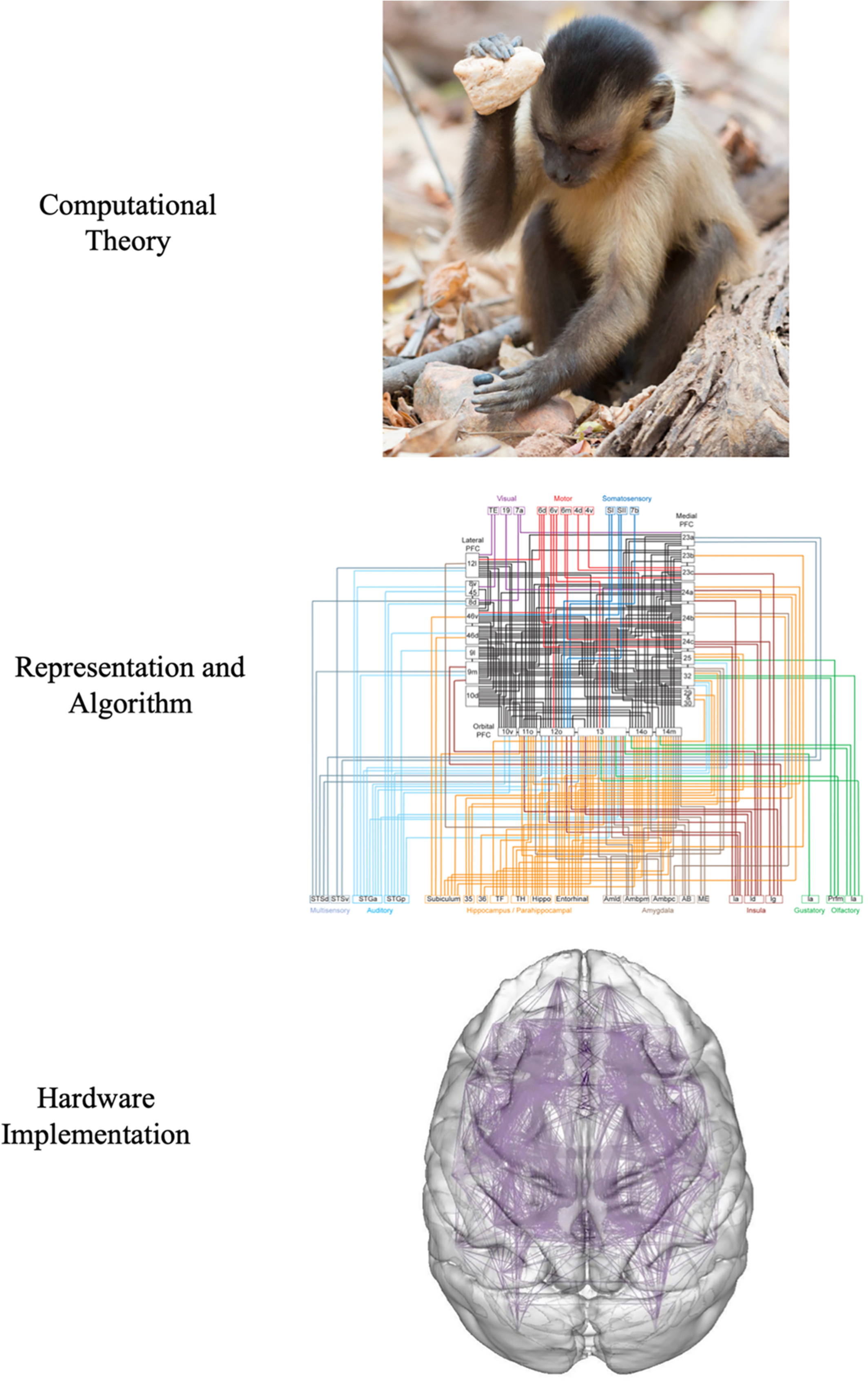
Computational perspective
- Computation
- Evolution, ethology, psychology
- Neuromechanics
- Representation & algorithm
- Somatotopy
- Hardware
- Muscles, tendons, spinal cord, brain
The cerebral symphony
Next time
- Perception
Resources
About
This talk was produced using Quarto, using the RStudio Integrated Development Environment (IDE), version 2026.1.0.392.
The source files are in R and R Markdown, then rendered to HTML using the revealJS framework. The HTML slides are hosted in a GitHub repo and served by GitHub pages: https://psu-psychology.github.io/psy-511-scan-fdns-2026-spring/
References
Footnotes
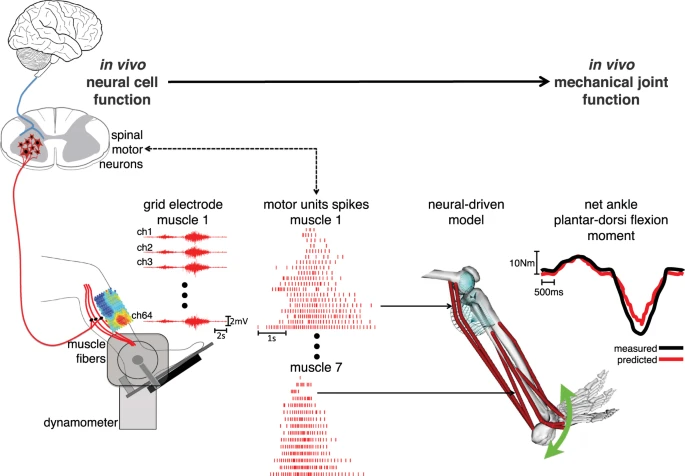
Linking between the neural and mechanical levels of motor function. Subjects perform a large repertoire of isometric ankle plantar-dorsi flexion contractions. High-density electromyograms are recorded and decomposed into the temporal events at which the underlying motor neurons discharged, thus opening a window into the central nervous system. This is done for seven major muscles spanning the ankle joint (see Methods Section). The decoded motor neuron discharges are directly used to control a subject-specific model of the musculoskeletal system. This enables reconstructing the net ankle moment over time, without knowledge of the experimental values (i.e. blinded validation) and without closed-loop feedback mechanism for compensating prediction errors (i.e. open-loop formulations). In this way, the predicted moment is purely contributed by motor neuron-controlled multi-muscle contractions acting on the skeletal system.
By CFCF - Own work, CC BY-SA 4.0, https://commons.wikimedia.org/w/index.php?curid=37700550
By CFCF - Own work, CC BY-SA 4.0, https://commons.wikimedia.org/w/index.php?curid=37700551
IIA and IIX are subtypes, see https://en.wikipedia.org/wiki/Skeletal_muscle
Remember afferents ascend (i.e, are inputs).
but not instantaneous
https://en.wikipedia.org/wiki/Primary_motor_cortex
Topographic map of the body.
By mailto:ralf@ark.in-berlin.de - File:Homunculus-ja.pngAfter Penfield and Rasmussen (1950), The Cerebral Cortex of Man.Modified from (an earlier version of) File:Homunculus-de.png by Was a bee., CC BY-SA 4.0, https://commons.wikimedia.org/w/index.php?curid=49377875
By I, Paskari, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=2344159
By OpenStax College - Anatomy & Physiology, Connexions Web site. http://cnx.org/content/col11496/1.6/, Jun 19, 2013., CC BY 3.0, https://commons.wikimedia.org/w/index.php?curid=30148015
By OpenStax College - Anatomy & Physiology, Connexions Web site. http://cnx.org/content/col11496/1.6/, Jun 19, 2013., CC BY 3.0, https://commons.wikimedia.org/w/index.php?curid=30148015
By BruceBlaus - Own work, CC BY-SA 4.0, https://commons.wikimedia.org/w/index.php?curid=46621399
By Sheryl Tan, Faull L, Curtis A - The Anatomical record, CC BY 4.0, https://commons.wikimedia.org/w/index.php?curid=152594765
By Grant, John Charles Boileau - An atlas of anatomy, / by regions 1962, Public Domain, https://commons.wikimedia.org/w/index.php?curid=30017222
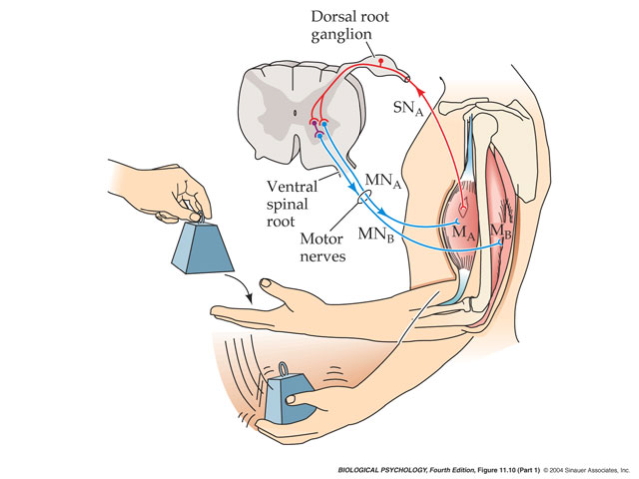
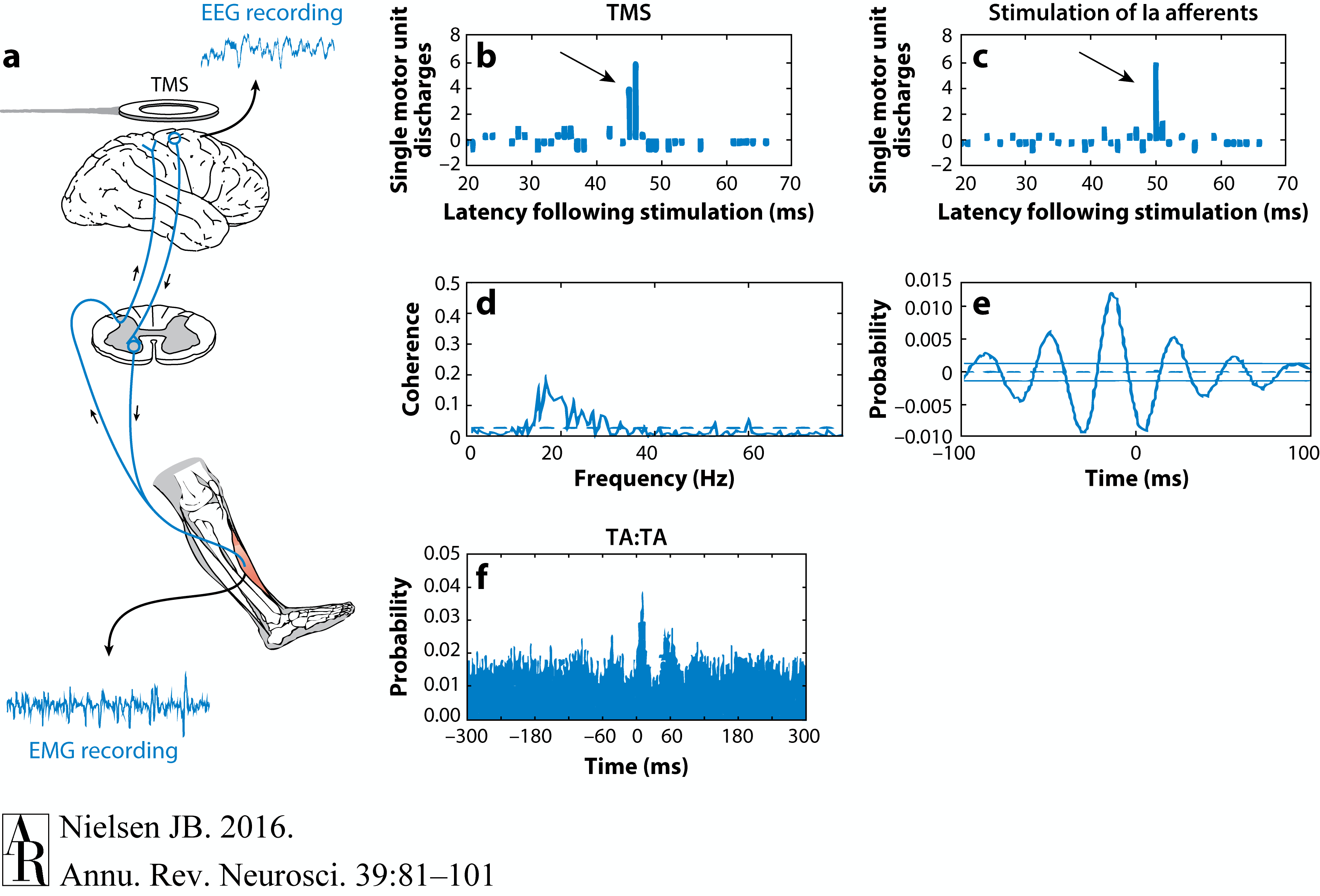
Evidence of corticomotoneuronal connections in human subjects. Indirect, noninvasive evidence of the existence of monosynaptic connections between corticospinal neurons and spinal motoneurons may be obtained in awake human subjects by transcranial magnetic stimulation (TMS) (b,c) and coherence analysis of either cortical [electroencephalogram (EEG)] and muscular activity [electromyogram (EMG)] (d,e) or two separate recordings of muscular activity (f). (b,c) Corticospinal neurons can be excited by a brief magnetic pulse applied by a magnetic coil placed over the appropriate part of the motor cortex in awake human subjects. If the intensity of the magnetic pulse is adjusted appropriately, the evoked descending volley in the corticospinal tract may elicit a subthreshold excitatory postsynaptic potential (EPSP) in the relevant spinal motoneurons. This EPSP may be demonstrated as a change in the discharge probability of a single motor unit recorded from the muscle (b). In the illustrated example, the subject was asked to voluntarily activate the tibialis anterior (TA) muscle, and the discharges of a single motor unit were recorded by a needle electrode inserted into the muscle. TMS elicited a short-lasting (2-ms) increase of discharge probability at a latency of 45 ms (b). The short duration of this peak is consistent with the short rise time of a monosynaptic EPSP. This interpretation is further supported by the observation that stimulation of Ia afferents with known monosynaptic connections to the motoneurons elicits a peak with a similar short duration (c). Data in panels b and c modified with permission from Nielsen & Petersen (1994). (d,e) EEG recorded from the motor cortex and EMG recorded from a voluntarily activated muscle (TA in the illustrated example) show rhythmic modulation of the recorded activity at a frequency of 15–35 Hz. As shown from a coherence analysis of the two signals in panel d, some of this activity is common for the two sites, suggesting a close link between cortical and muscular activity. Panel e shows the EEG and EMG activities are not always synchronous but may show a time lag, which is in the range expected for a fast-conducting direct pathway to the motoneurons. Data in panels d and e modified with permission from Hansen et al. (2002). (f) A monosynaptic origin of corticomuscular coherence is further supported by the observation of short-term synchrony between the discharges of pairs of TA motor units, which may be related to the coherence in the 15–35-Hz frequency band. The subject was asked to voluntarily activate the TA muscle, and the discharges of two different TA motor units were recorded with needle electrodes. The short duration of the central peak of synchronization suggests that the motor unit activities are modulated by a common (monosynaptic) input from collaterals of last-order neurons, which are in all likelihood identical to corticomotoneuronal cells. The secondary peaks at lags of approximately 50–60 ms on either side of the central peak suggest that this last-order input modulates the discharge of the motor units at a frequency of about 20–30 Hz, i.e., corresponding to the coherence observed in the paired EEG-EMG recordings in panels b and c. Data in panel f modified with permission from Nielsen & Kagamihara (1994).
Nishikawa et al. (2007)
By Rhcastilhos. And Jmarchn. - Schematic_diagram_of_the_human_eye_with_English_annotations.svg, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=1597930
By Vanessa Ezekowitz - Hand-drawn based on File:AcuityHumanEye.jpg by Hans-Werner Hunziker, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=7327065
By .Koen - Own work based on: Simple vestibulo-ocular reflex.PNG and Image:ThreeNeuronArc.png, CC BY-SA 4.0, https://commons.wikimedia.org/w/index.php?curid=4130081
By Student BSMU, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=3004633
By Greyson Orlando - Own work, Public Domain, https://commons.wikimedia.org/w/index.php?curid=1451923
By OpenStax College - Anatomy & Physiology, Connexions Web site. http://cnx.org/content/col11496/1.6/, Jun 19, 2013., CC BY 3.0, https://commons.wikimedia.org/w/index.php?curid=30147999
Cranial nerve
Animal movement, as represented by a neuromechanical system diagram. Whether in humans or moths, neurons activate muscles by transmitting electrical impulses over time (t)—frequently digital, all-or-nothing events termed action potentials or spikes. Muscles transform the time-varying neural motor commands u(t) into mechanical forces f(t). Muscles actuate the body, but it is the interaction of the body with the surrounding environment that produces locomotion y(t). Meanwhile, myriad sensors detect not only the body and how it interacts with environment, but also internal states of the animal. All that information feeds back through the animal’s nervous system. Sensor signals can be combined with reference signals, illustrated by red arrows. In this schematic, r1(t) represents a brain-directed task such as “fly at a steady speed,” and r2(t) represents exogenous sensory cues such as a moth might receive when tracking the movement of a flower
Figure 1. The G-SLIP model: (a) its four transformations using model parameter adjustment: SLIP, TSL, SLIP-RF, and R-SLIP model. (b) Running of the G-SLIP model, which is composed of alternating stance phase and flight phase in each stride.
https://www.youtube.com/@mendip89/shorts
Figure 1 | Models of the spinal neuronal circuit that controls locomotion. a, Conventional spinal locomotor models are built from pools of motor and other spinal neurons. Each pool excites one of a pair of muscles — the flexor or extensor muscle — while inhibiting the activity of the other pool. This mutual inhibition means that neuronal activity (indicated for each neuron by vertical lines along a colour-coded timeline) alternates back and forth between the pools, including in the flexor and extensor motor neurons, which drive alternating muscle movements to create steps (or other types of movement). b, Lindén et al.2 propose a different model in which the collective activity of a near-randomly connected pool of neurons covers all of the phases of one bout of movement. Each neuron contributes to this cyclical pattern of activity, with the flexor and extensor motor neurons still alternating as part of the cycle, driving muscle movement.
By created by User:ish shwar (original .png deleted), .svg by Rohieb - sagittal section image based on Minifie et al. (1973:); articulation places are from Catford (1977), CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=1771968
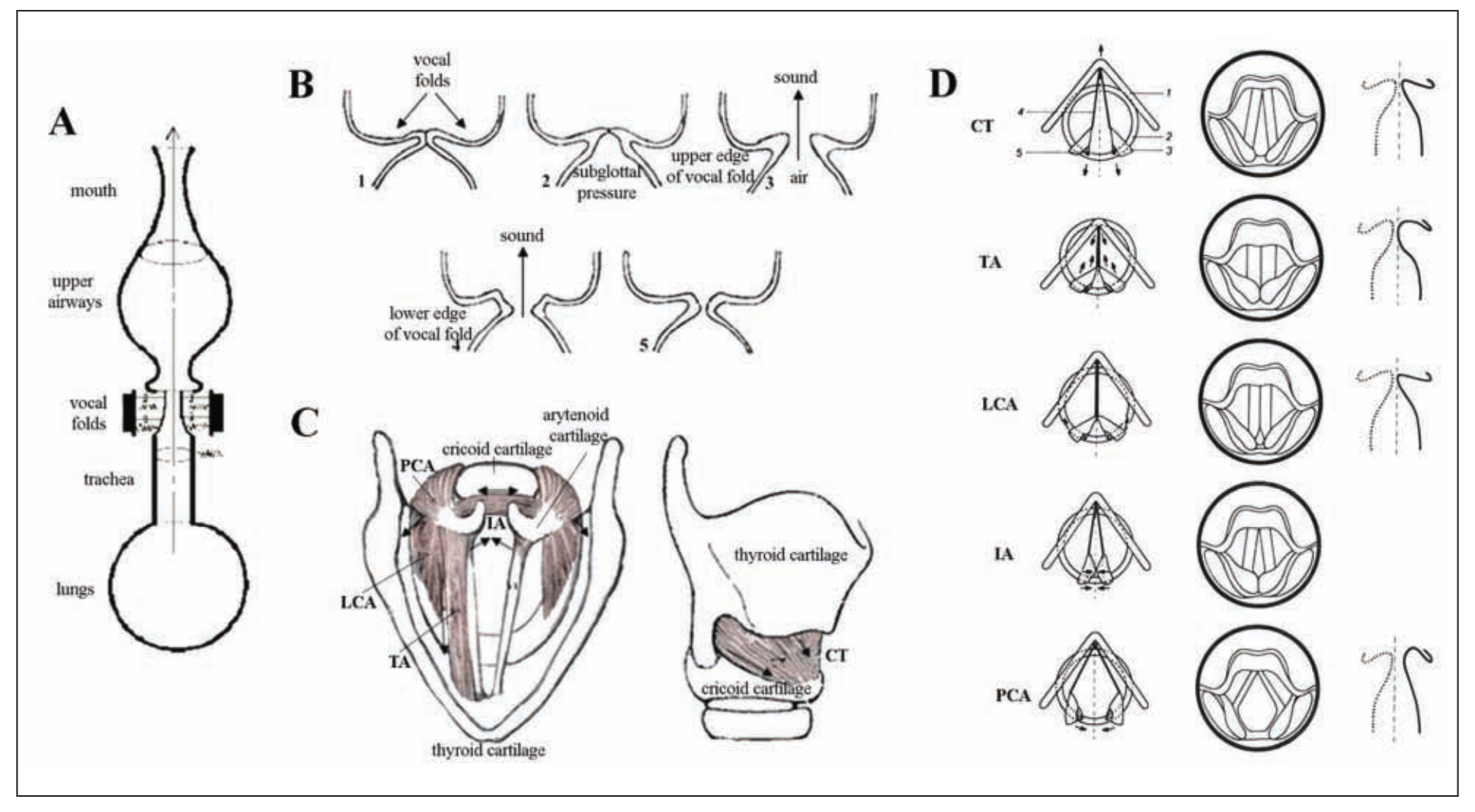
Figure 1. (A) Schematic view of the vocal and respiratory tracts. Voice originates in the larynx. First, the expiratory airflow from the lungs reaches the larynx through the trachea, where it sets the closed vocal fold tissue into self-excited oscillatations, due to which the larynx becomes the source of voice sound. Further, pressure from the vocal fold oscillations is resonated through the vocal tract and radiated from the mouth as voice. (B) Schematic sequence of events preceding voice production: 1) The vocal folds close immediately prior to voice production; 2) subglottal air pressure builds up below the vocal folds during exhalation; 3) the lower and upper edge of the vocal folds separate subsequently with the release of air and sound generation; 4) the vocal folds reapproximate, starting from their lower edge; and 5) the vocal folds close completely before the next sound production.(C) Superior and lateral views of the human larynx. Intrinsic laryngeal muscles and cartilages. TA = thyroarytenoid muscle; LCA = lateral cricoarytenoid muscle; PCA = posterior cricoarytenoid muscle; IA = interarytenoid muscle; CT = cricothyroid muscle. The arrows show the directions of the muscle contractions. (D) Schematic presentation of the laryngeal muscle function. The left column shows the location of the cartilages and the edge of the vocal folds when each of the laryngeal muscles is active. The arrows indicate the directions of the force exerted: 1) thyroid cartilage, 2) cricoid cartilage, 3) arytenoid cartilages, 4) vocal ligament, and 5) posterior cricoarytenoid ligament. The middle column shows the laryngeal view. The right column shows contours of the frontal section at the middle of the membranous portion of the vocal fold. The dotted line shows a state in which no muscle is activated. Reprinted from Hirano, copyright 1981, with kind permission of Springer Science+Business Media.
Full caption: The cerebral networks supporting primate-general (gray arrows) and human-specific (black) aspects of vocal communication are assumed to be closely intertwined at the level of the basal ganglia. Dashed lines indicate that the basal ganglia motor loop undergoes a dynamic ontogenetic reorganization during spoken language acquisition in that a left-hemisphere cortical storage site of syllable-sized motor programs gradually emerges. Amygdala etc., Amygdala and other structures of the limbic system; ACC, anterior cingulate cortex; SMA, supplementary motor area; GPi, internal segment of globus pallidus; SNr/SNc, substantia nigra, pars reticulata/pars compacta; PAG, periaqueductal gray; vCPG, vocal central pattern generator. Data from Ackermann et al. (2014). B, Gestural architecture of the word “speaking.” Laryngeal activity (bottom line) is a crucial part of the respective movement sequence and must be adjusted to other vocal tract excursions. Articulatory gestures are assorted into syllabic units; gesture bundles pertaining to strong and weak syllables are rhythmically patterned to form metrical feet. Data from Ziegler (2010).

PSY 511.001 | © 2025 Rick Gilmore under CC BY 4.0