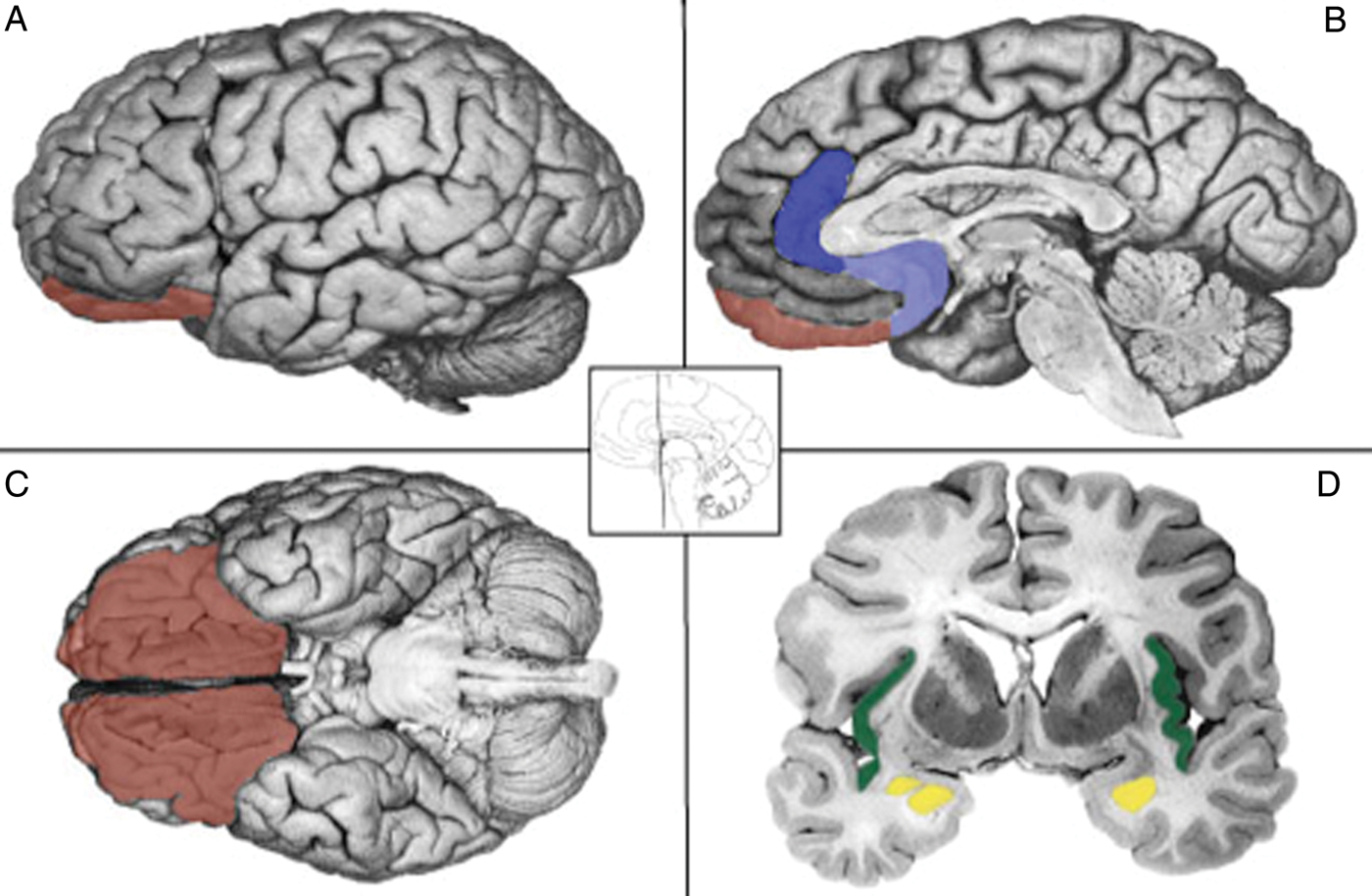
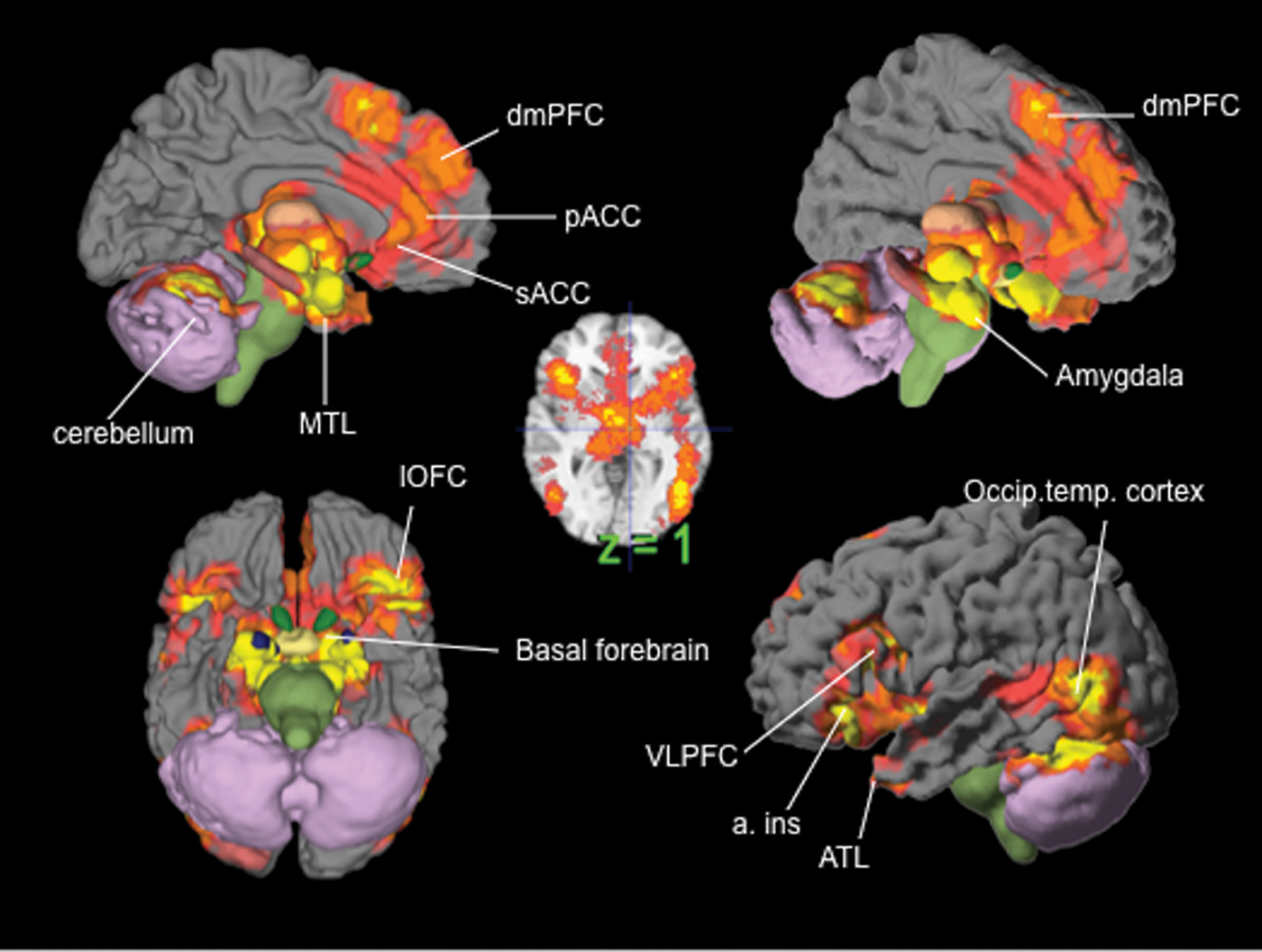
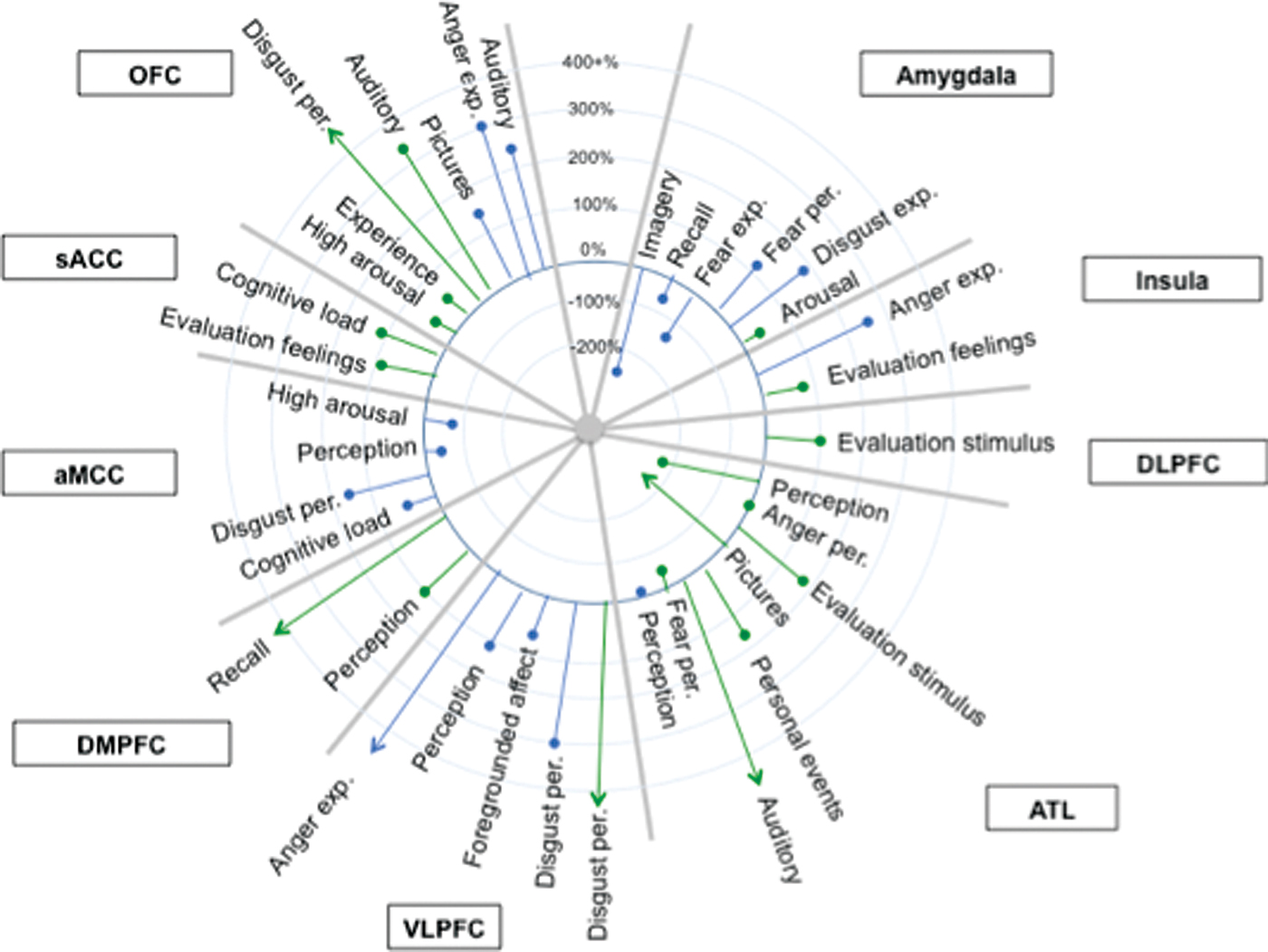
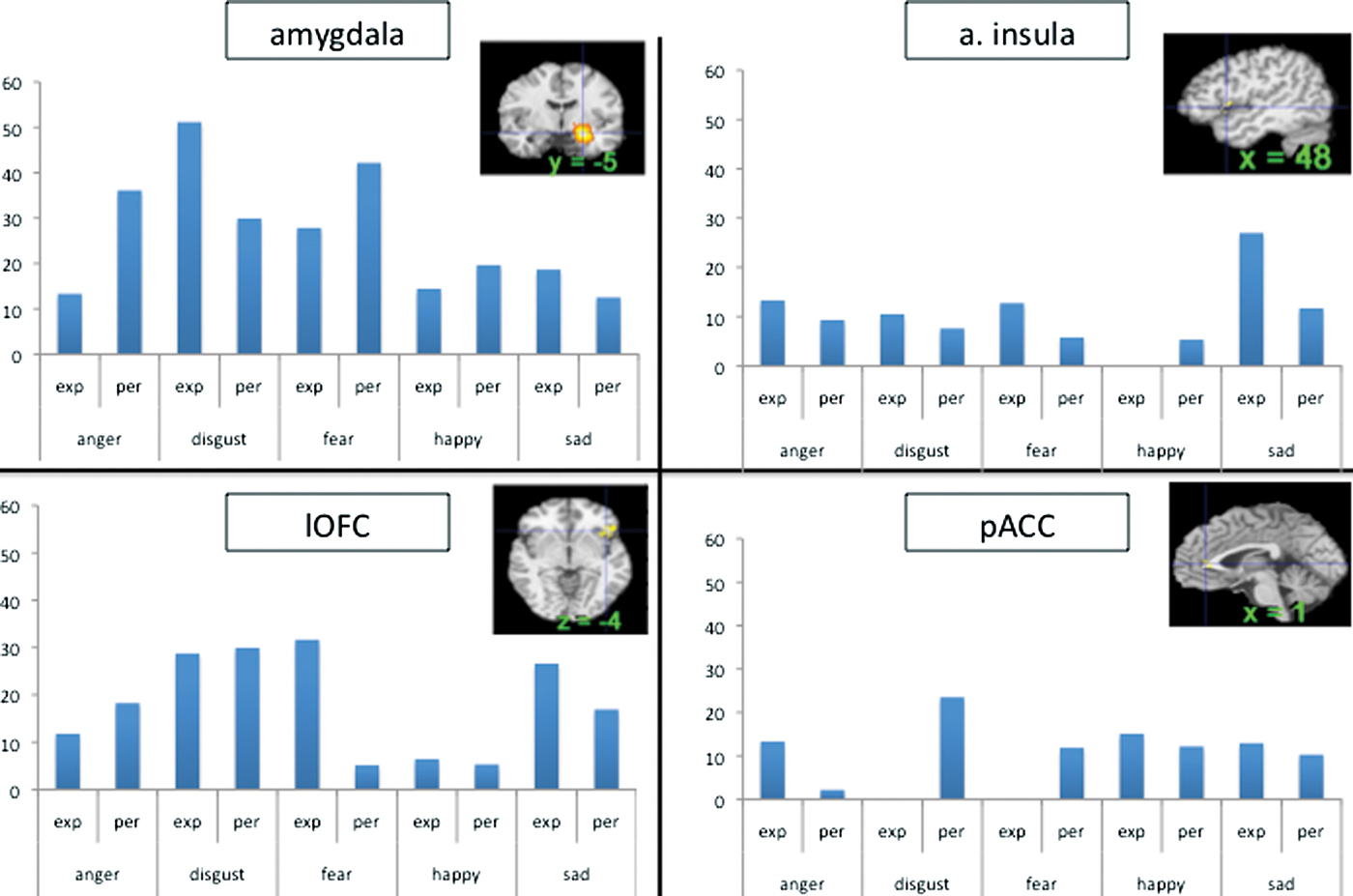
ashikkerib. (2007, December). Brain mechanisms of pleasure and addiction. You
Tube. Retrieved from
https://www.youtube.com/watch?v=de_b7k9kQp0
Barrett, L. F. (2017). The theory of constructed emotion: An active inference account of interoception and categorization.
Social Cognitive and Affective Neuroscience,
12, 1–23.
https://doi.org/10.1093/scan/nsw154
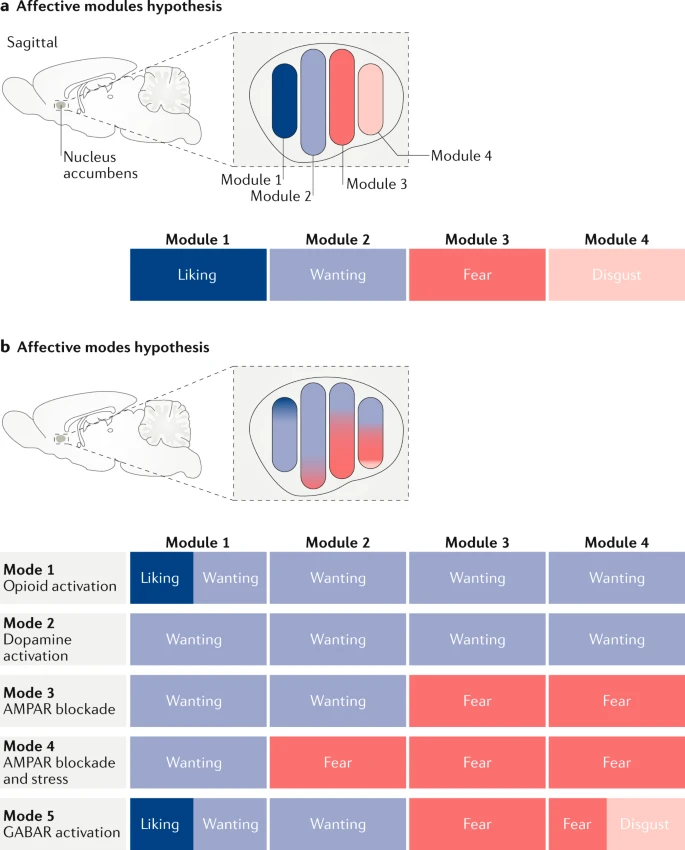
Berridge, K. C. (2019). Affective valence in the brain: Modules or modes?
Nature Reviews. Neuroscience,
20, 225–234.
https://doi.org/10.1038/s41583-019-0122-8
Brandão, M. L., Zanoveli, J. M., Ruiz-Martinez, R. C., Oliveira, L. C., & Landeira-Fernandez, J. (2008). Different patterns of freezing behavior organized in the periaqueductal gray of rats: Association with different types of anxiety.
Behavioural Brain Research,
188(1), 1–13.
https://doi.org/10.1016/j.bbr.2007.10.018
Caravaca, F., Cuevas, Á., & Cuevas, R. (2025). From prompts to power: Measuring the energy footprint of
LLM inference.
arXiv [Cs.AI].
https://doi.org/10.48550/arXiv.2511.05597
Clapp, P., Bhave, S. V., & Hoffman, P. L. (n.d.). How
Adaptation of the
Brain to
Alcohol
Leads to
Dependence. Retrieved from
http://pubs.niaaa.nih.gov/publications/arh314/310-339.htm
daleswartzentruber. (2007, October). Conditioned suppression of a rat’s lever pressing. You
Tube. Retrieved from
https://www.youtube.com/watch?v=ZlZekx1P1g4
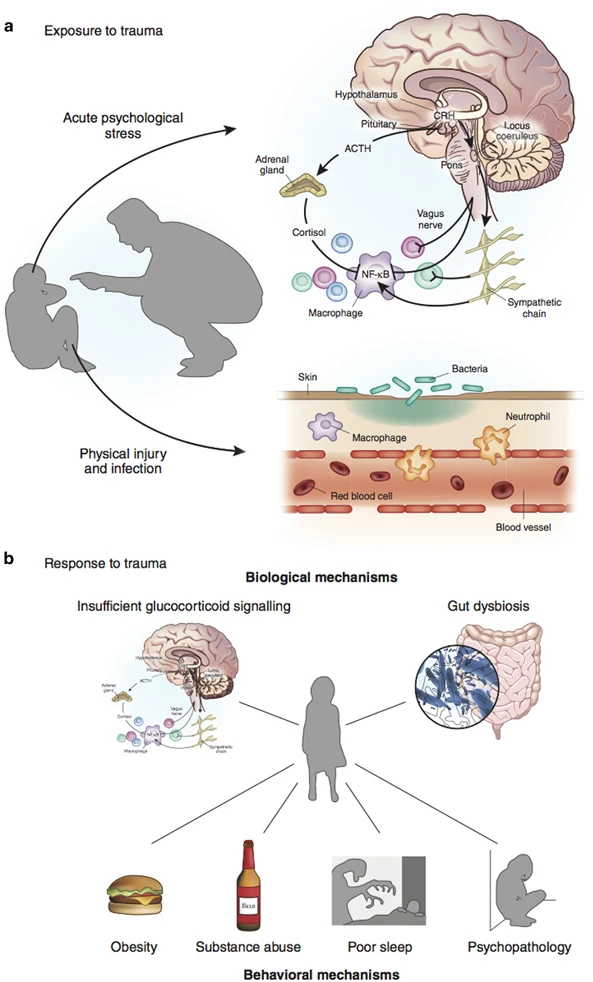
Danese, A., & J Lewis, S. (2017). Psychoneuroimmunology of early-life stress: The hidden wounds of childhood trauma?
Neuropsychopharmacology: Official Publication of the American College of Neuropsychopharmacology,
42, 99–114.
https://doi.org/10.1038/npp.2016.198
Darwin, C. (1899).
The expression of emotion in man and animals. New York: Project Gutenberg. Retrieved from
https://www.gutenberg.org/files/1227/1227-h/1227-h.htm
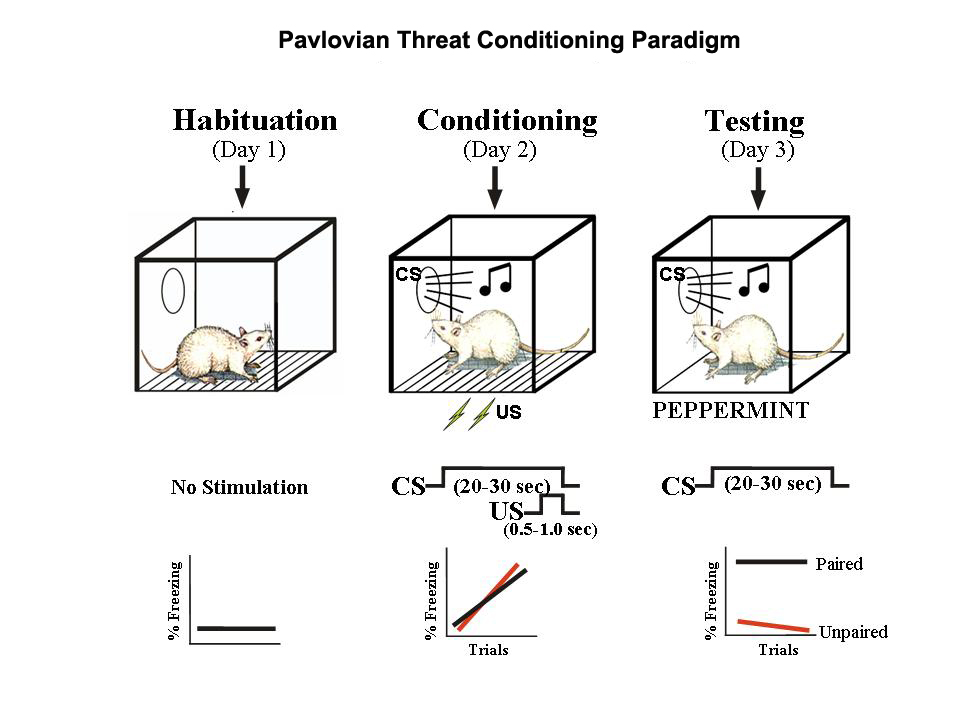
Davis, M. (1992). The role of the amygdala in fear-potentiated startle: Implications for animal models of anxiety.
Trends in Pharmacological Sciences,
13, 35–41.
https://doi.org/10.1016/0165-6147(92)90014-W
Deussing, J. M., & Chen, A. (2018). The
Corticotropin-Releasing factor family: Physiology of the stress response.
Physiological Reviews,
98(4), 2225–2286.
https://doi.org/10.1152/physrev.00042.2017
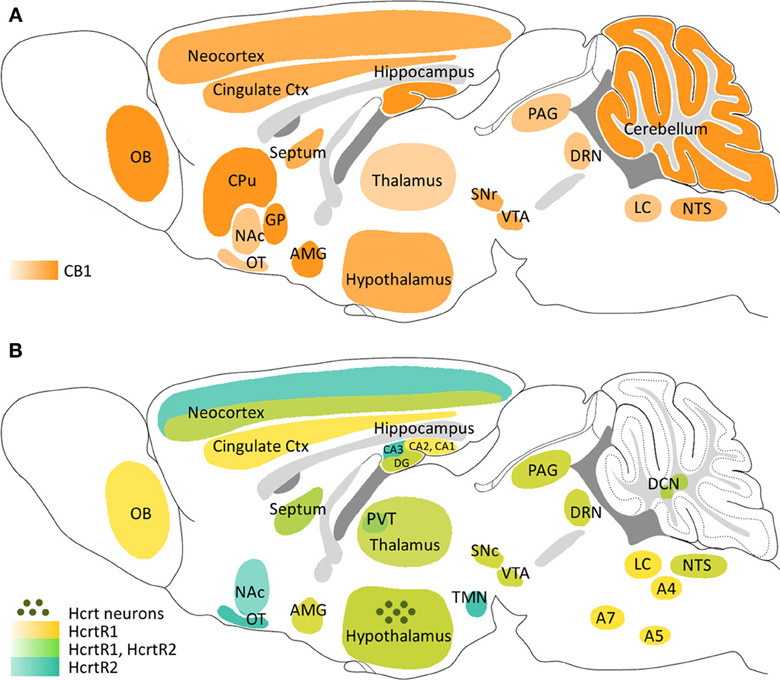
Flores, Á., Maldonado, R., & Berrendero, F. (2013). Cannabinoid-hypocretin cross-talk in the central nervous system: What we know so far.
Neuropharmacology,
7, 256.
https://doi.org/10.3389/fnins.2013.00256
Glerean, E. (n.d.). Embody. Retrieved February 26, 2026, from
https://version.aalto.fi/gitlab/eglerean/embody
Harrison, N. A., Gray, M. A., Gianaros, P. J., & Critchley, H. D. (2010). The embodiment of emotional feelings in the brain.
J. Neurosci.,
30(38), 12878–12884.
https://doi.org/10.1523/JNEUROSCI.1725-10.2010
Heath, R. G. (1963). Electrical self-stimulation of the brain in man.
American Journal of Psychiatry,
120(6), 571–577.
https://doi.org/10.1176/ajp.120.6.571
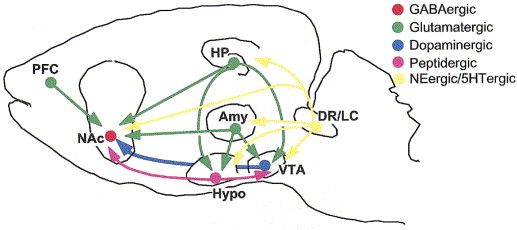
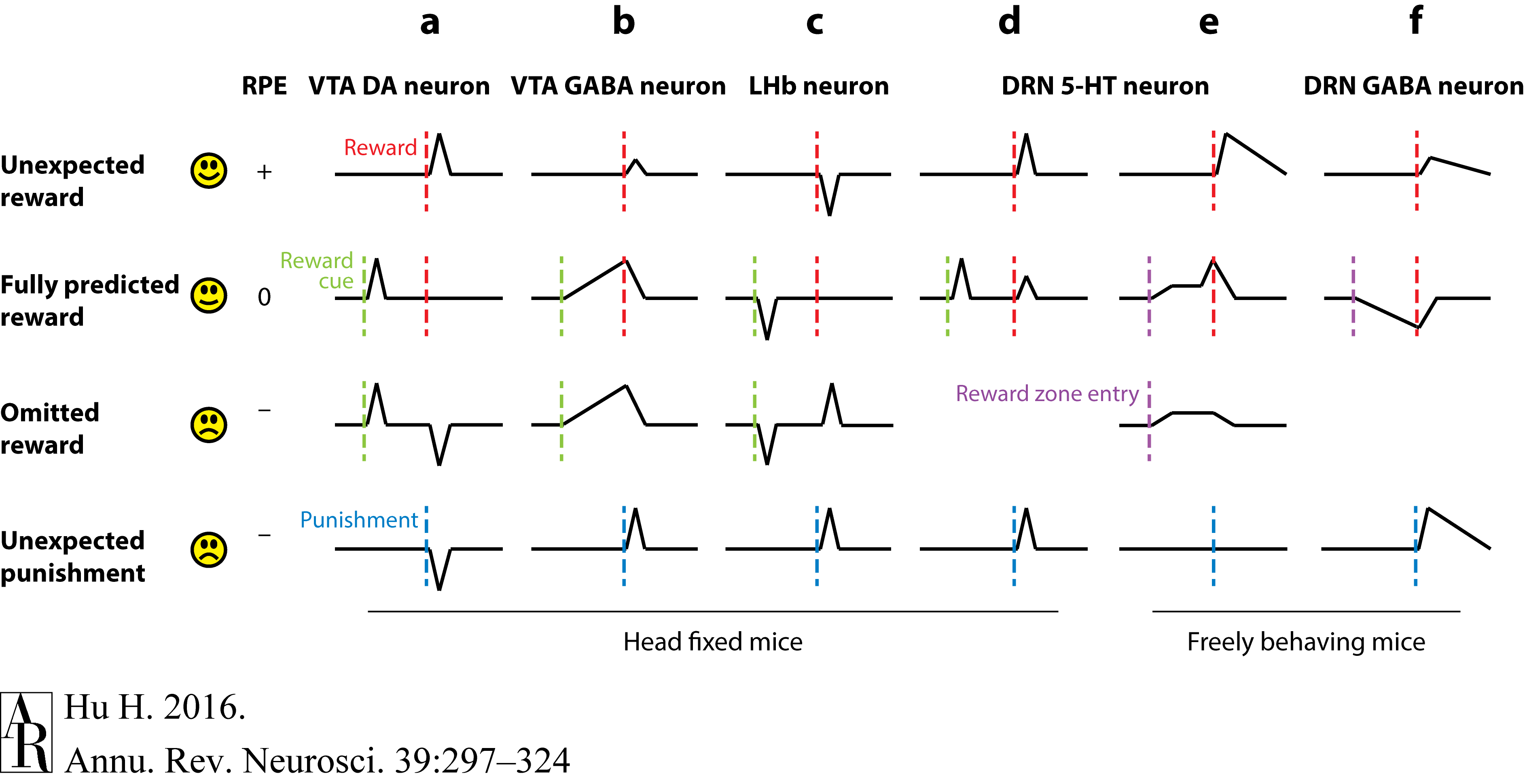
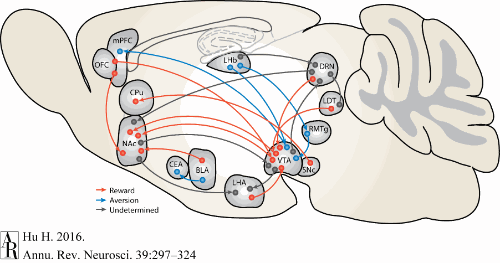
Hu, H. (2016). Reward and aversion.
Annual Review of Neuroscience,
39, 297–324.
https://doi.org/10.1146/annurev-neuro-070815-014106
Kadmiel, M., & Cidlowski, J. A. (2013). Glucocorticoid receptor signaling in health and disease.
Trends in Pharmacological Sciences,
34(9), 518–530.
https://doi.org/10.1016/j.tips.2013.07.003
Kohls, G., Chevallier, C., Troiani, V., & Schultz, R. T. (2012). Social ‘wanting’dysfunction in autism: Neurobiological underpinnings and treatment implications.
Journal of Neurodevelopmental Disorders,
4(10), 1–20.
https://doi.org/10.1186/1866-1955-4-10
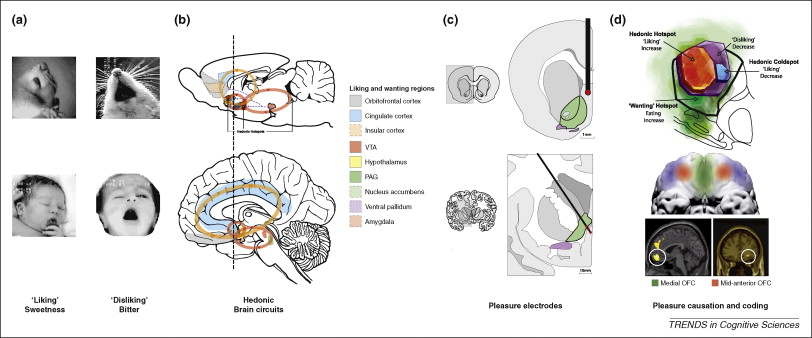
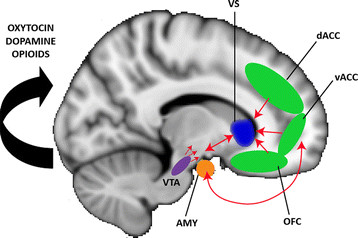
Kringelbach, M. L., & Berridge, K. C. (2009). Towards a functional neuroanatomy of pleasure and happiness. Trends in Cognitive Sciences, 13(11), 479–487.
Lindquist, K. A., Wager, T. D., Kober, H., Bliss-Moreau, E., & Barrett, L. F. (2012). The brain basis of emotion: A meta-analytic review.
Behav. Brain Sci.,
35(3), 121–143.
https://doi.org/10.1017/S0140525X11000446
Malezieux, M., Klein, A. S., & Gogolla, N. (2023). Neural circuits for emotion.
Annual Review of Neuroscience,
46, 211–231.
https://doi.org/10.1146/annurev-neuro-111020-103314
Medina, J. F., Repa, J. C., Mauk, M. D., & LeDoux, J. E. (2002). Parallels between cerebellum-and amygdala-dependent conditioning.
Nature Reviews Neuroscience,
3(2), 122–131.
https://doi.org/10.1038/nrn728
Mesquita, B., & Frijda, N. H. (1992). Cultural variations in emotions: A review.
Psychological Bulletin,
112, 179–204.
https://doi.org/10.1037/0033-2909.112.2.179
Milner, P. M. (1989). The discovery of self-stimulation and other stories.
Neuroscience & Biobehavioral Reviews,
13(2–3), 61–67.
https://doi.org/10.1016/S0149-7634(89)80013-2
Musazzi, L., Tornese, P., Sala, N., & Popoli, M. (2017). Acute or chronic? A stressful question.
Trends in Neurosciences.
https://doi.org/10.1016/j.tins.2017.07.002
Nestler, E. J., & Carlezon, W. A. (2006). The mesolimbic dopamine reward circuit in depression.
Biological Psychiatry,
59(12), 1151–1159.
https://doi.org/10.1016/j.biopsych.2005.09.018
Nummenmaa, L., Glerean, E., Hari, R., & Hietanen, J. K. (2014). Bodily maps of emotions.
Proceedings of the National Academy of Sciences of the United States of America,
111(2), 646–651.
https://doi.org/10.1073/pnas.1321664111
O’Donnell, J. (2025, May 20). We did the math on
AI’s energy footprint. Here’s the story you haven’t heard. Retrieved February 25, 2026, from
https://www.technologyreview.com/2025/05/20/1116327/ai-energy-usage-climate-footprint-big-tech/
Pellman, B. A., & Kim, J. J. (2016). What can ethobehavioral studies tell us about the brain’s fear system?
Trends in Neurosciences,
39(6), 420–431.
https://doi.org/10.1016/j.tins.2016.04.001
Pixar. (2014, December). Inside out - official
US trailer. Youtube. Retrieved from
https://www.youtube.com/watch?v=yRUAzGQ3nSY
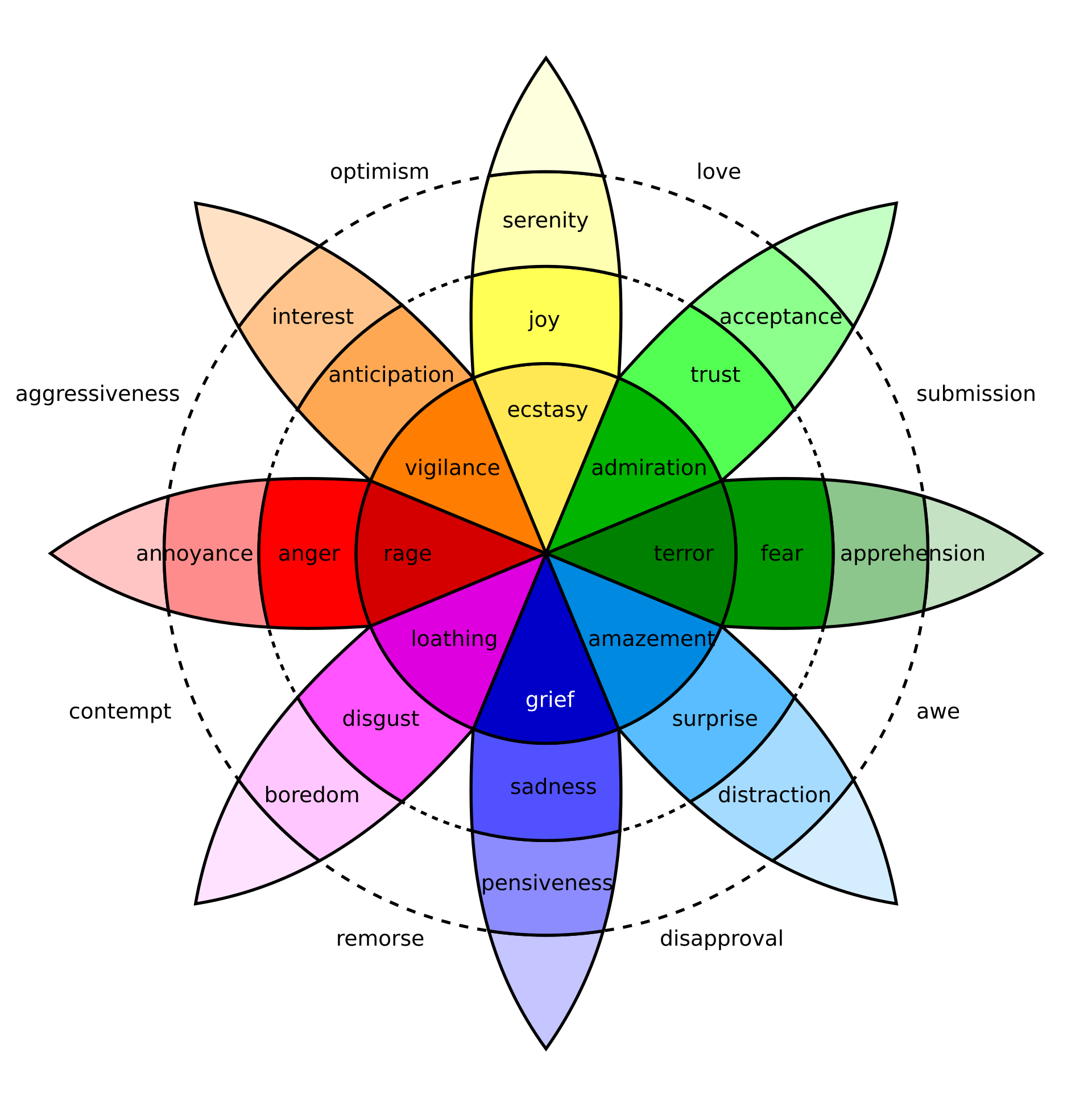
"Robert Plutchik". (2004, October 11). Robert plutchik. Retrieved February 18, 2026, from
https://en.wikipedia.org/wiki/Robert_Plutchik
Sapolsky, R. (1994). Why zebras don’t get ulcers. New York, NY: W.H. Freeman.
Shubhamku. (2025, July 31). The environmental footprint of large language models: A looming challenge for
AI. Retrieved February 25, 2026, from
https://medium.com/@shubhamku2022/the-environmental-footprint-of-large-language-models-a-looming-challenge-for-ai-40cd8d0e6452
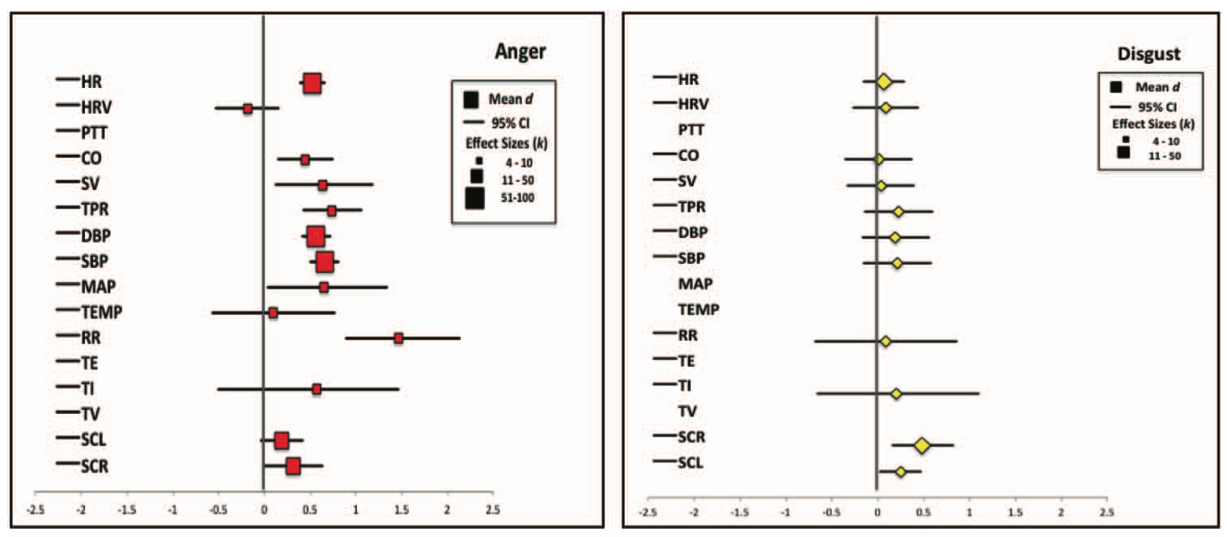
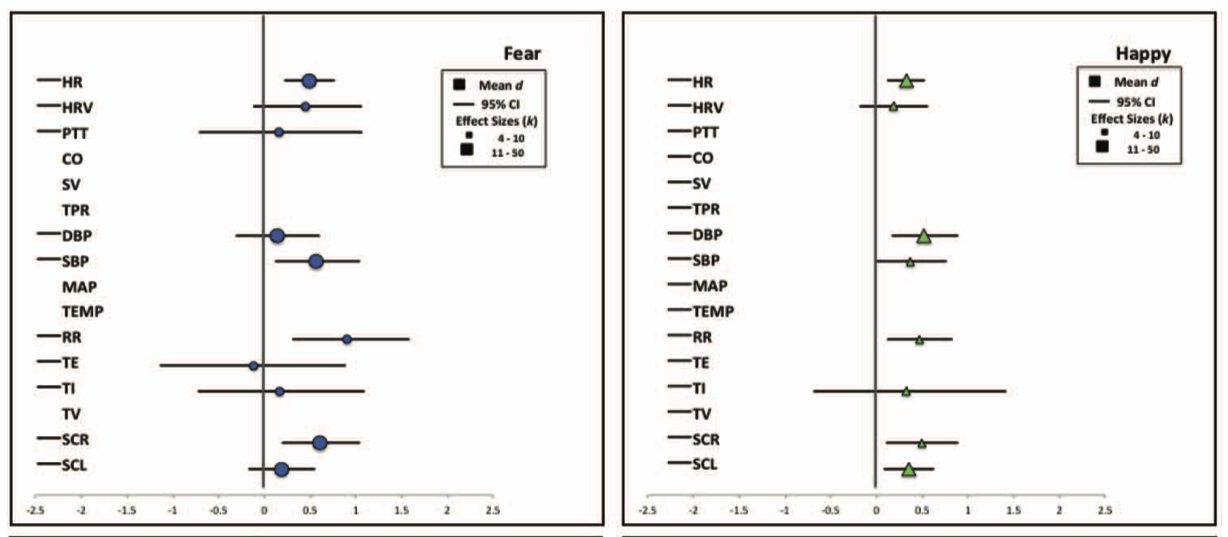
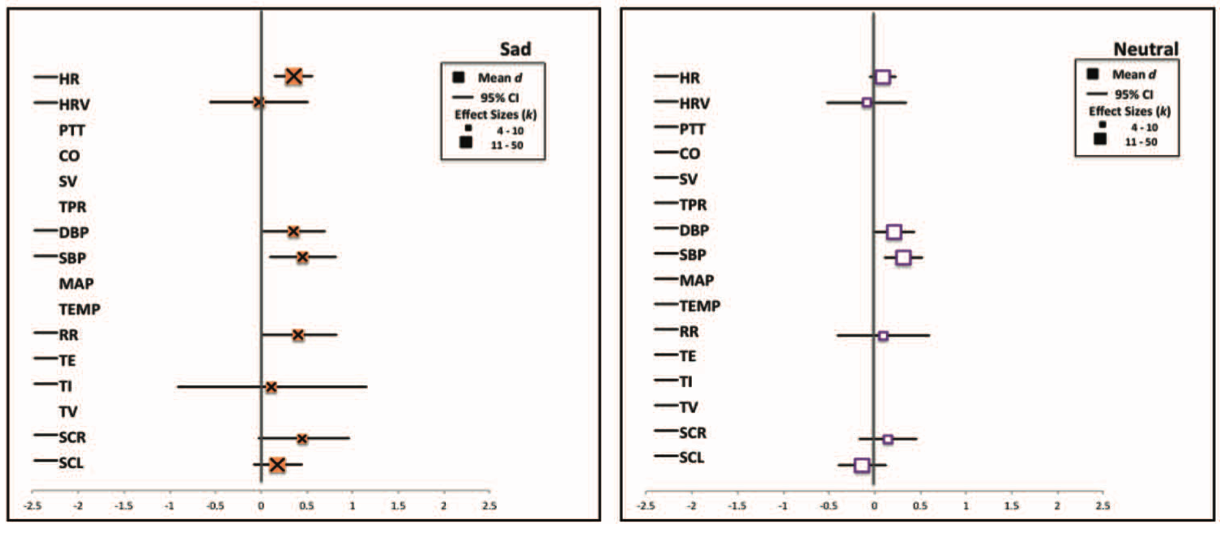
Siegel, E. H., Sands, M. K., Van den Noortgate, W., Condon, P., Chang, Y., Dy, J., … Barrett, L. F. (2018). Emotion fingerprints or emotion populations? A meta-analytic investigation of autonomic features of emotion categories.
Psychological Bulletin,
144(4), 343–393.
https://doi.org/10.1037/bul0000128
The Ed Sullivan Show. (2020).
Smokey robinson & the miracles "I second that emotion, if you can want, going to a go-go". You
Tube. Retrieved from
https://www.youtube.com/watch?v=2ia6zQLOPfU&list=RD2ia6zQLOPfU&start_radio=1
Tremblay, P., & Dick, A. S. (2016). Broca and
Wernicke are dead, or moving past the classic model of language neurobiology.
Brain and Language,
162, 60–71.
https://doi.org/10.1016/j.bandl.2016.08.004
Wager, T. D., Kang, J., Johnson, T. D., Nichols, T. E., Satpute, A. B., & Barrett, L. F. (2015). A bayesian model of category-specific emotional brain responses.
PLoS Computational Biology,
11, e1004066.
https://doi.org/10.1371/journal.pcbi.1004066
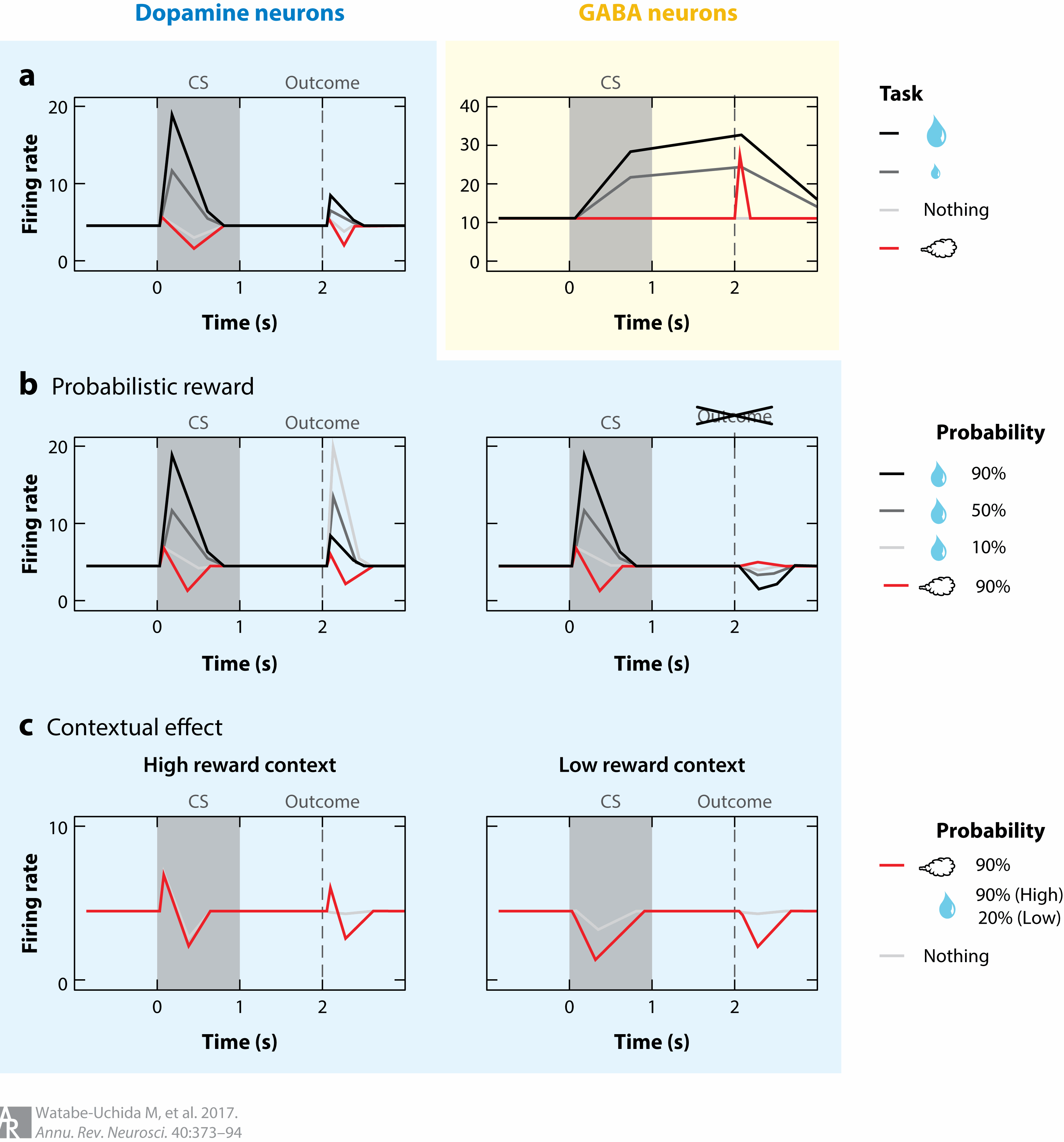
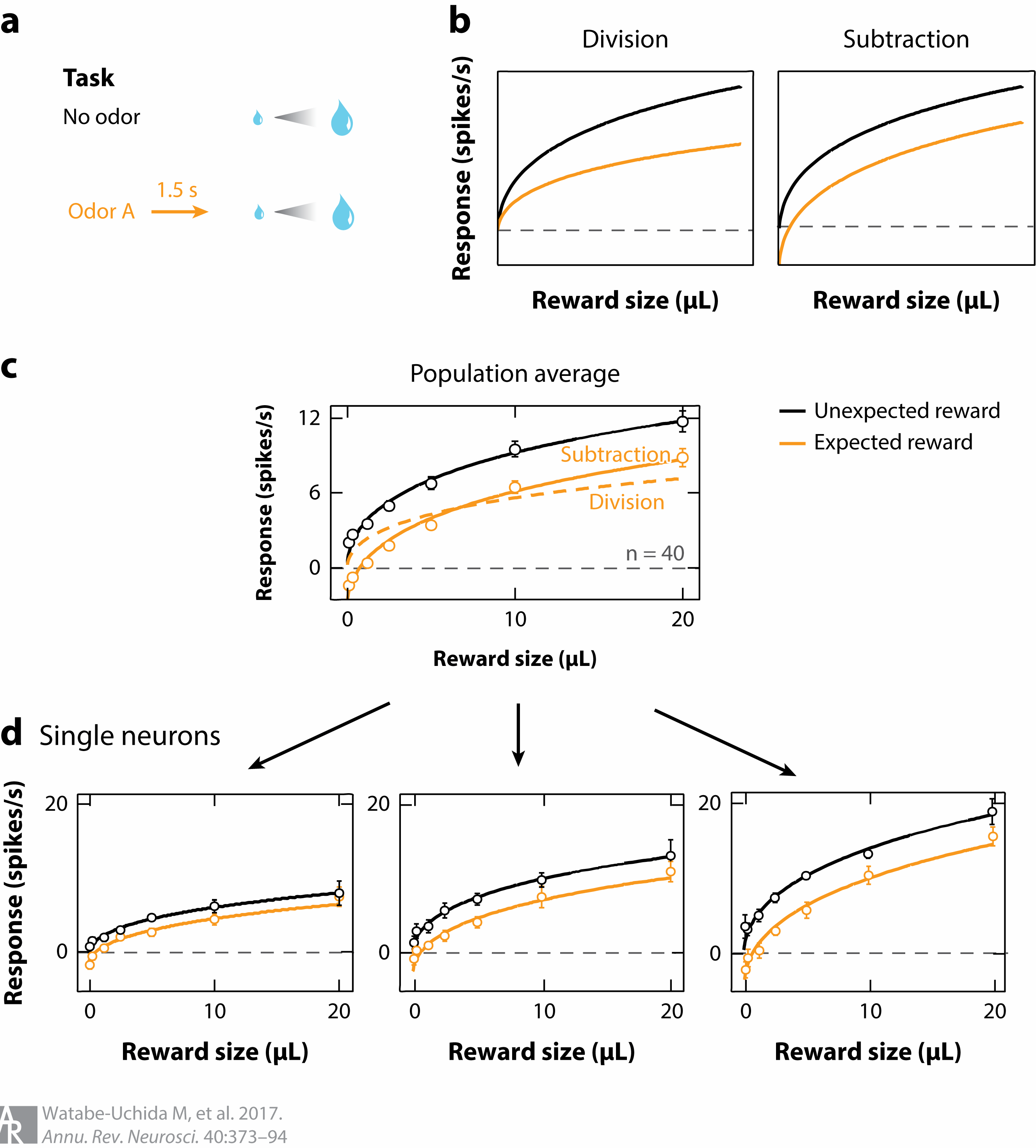
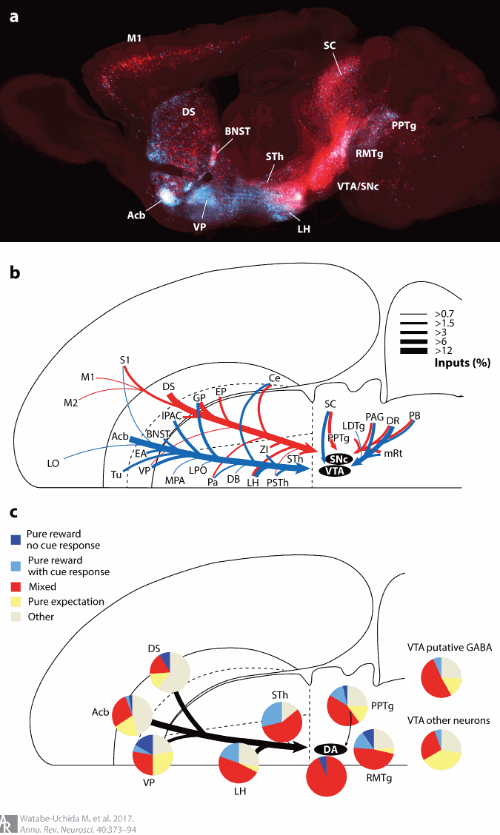
Watabe-Uchida, M., Eshel, N., & Uchida, N. (2017). Neural circuitry of reward prediction error.
Annual Review of Neuroscience,
40, 373–394.
https://doi.org/10.1146/annurev-neuro-072116-031109
Wikipedia contributors. (2025a, September 9).
James–Lange theory. Retrieved from
https://en.wikipedia.org/wiki/James%E2%80%93Lange_theory
Wikipedia contributors. (2025b, September 27).
Cannon–Bard theory. Retrieved from
https://en.wikipedia.org/wiki/Cannon%E2%80%93Bard_theory
Wikipedia contributors. (2025c, October 3). Semantic network. Retrieved from
https://en.wikipedia.org/wiki/Semantic_network
Wikipedia contributors. (2025d, November 5). Two-factor theory of emotion. Retrieved from
https://en.wikipedia.org/wiki/Two-factor_theory_of_emotion