Anderson, C. M., & Swanson, R. A. (2000). Astrocyte glutamate transport: Review of properties, regulation, and physiological functions.
Glia,
32(1), 1–14.
https://doi.org/10.1002/1098-1136(200010)32:1<1::AID-GLIA10>3.0.CO;2-W
Eyherabide, H. G., Rokem, A., Herz, A. V. M., Samengo, I., Eyherabide, H. G., Rokem, A., … Samengo, I. (2009). Bursts generate a non-reducible spike-pattern code.
Frontiers in Neuroscience,
3, 1.
https://doi.org/10.3389/neuro.01.002.2009
Hastoy, B., Clark, A., Rorsman, P., & Lang, J. (2017). Fusion pore in exocytosis: More than an exit gate? A
\(\beta\)-cell perspective.
Cell Calcium,
68, 45–61.
https://doi.org/10.1016/j.ceca.2017.10.005
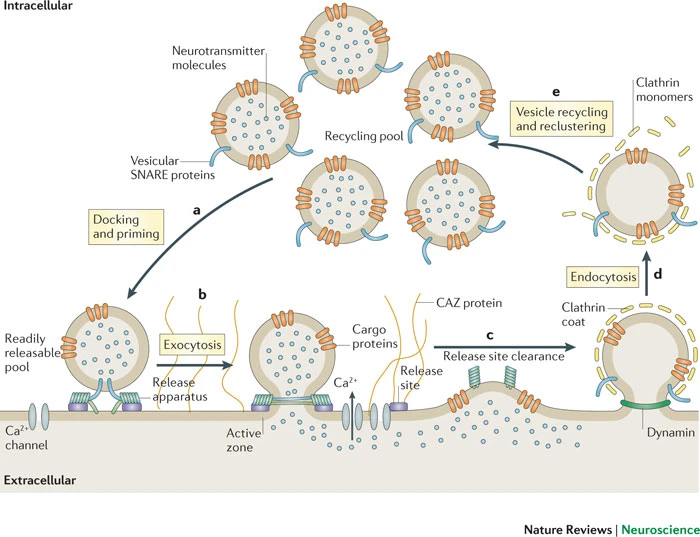
Haucke, V., Neher, E., & Sigrist, S. J. (2011). Protein scaffolds in the coupling of synaptic exocytosis and endocytosis.
Nature Reviews. Neuroscience,
12(3), 127–138.
https://doi.org/10.1038/nrn2948
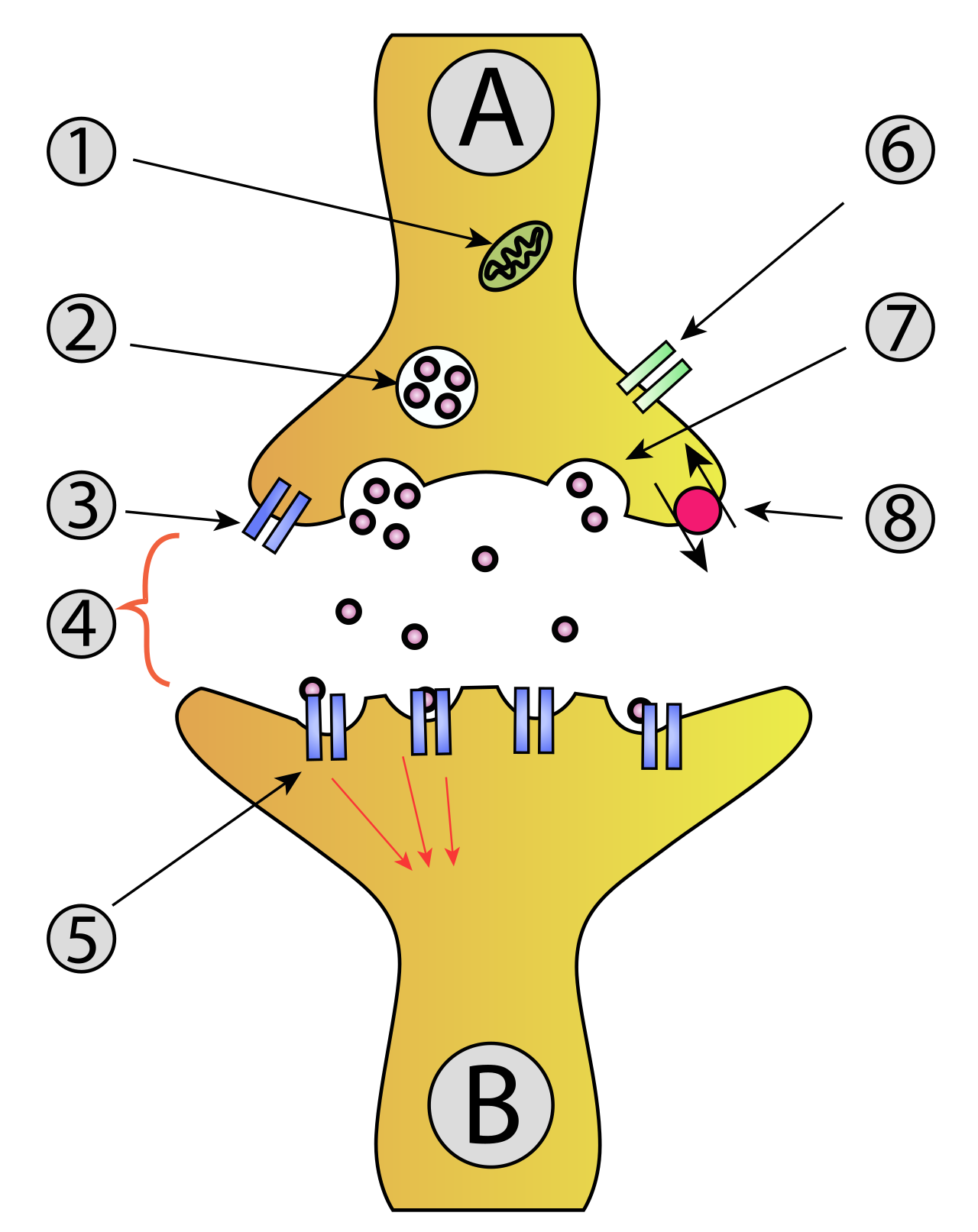
Henley, C. (2021). Synapse structure. In
Foundations of neuroscience. Michigan State University Libraries. Retrieved from
https://openbooks.lib.msu.edu/neuroscience/chapter/synapse-structure/
Musikladen. (2020).
Stevie Wonder - Superstition (1974). You
Tube. Retrieved from
https://www.youtube.com/watch?v=97hwNY3ni10
Wikipedia contributors. (2025a, February 7). Inhibitory postsynaptic potential. Retrieved from
https://en.wikipedia.org/wiki/Inhibitory_postsynaptic_potential
Wikipedia contributors. (2025b, July 19). Excitatory postsynaptic potential. Retrieved from
https://en.wikipedia.org/wiki/Excitatory_postsynaptic_potential
Wikipedia contributors. (2025c, August 30). Nerve conduction velocity. Retrieved from
https://en.wikipedia.org/wiki/Nerve_conduction_velocity
Wikipedia contributors. (2025d, October 27). Ligand-gated ion channel. Retrieved from
https://en.wikipedia.org/wiki/Ligand-gated_ion_channel
Wikipedia contributors. (2026a, March 19). Synaptic vesicle. Retrieved from
https://en.wikipedia.org/wiki/Synaptic_vesicle
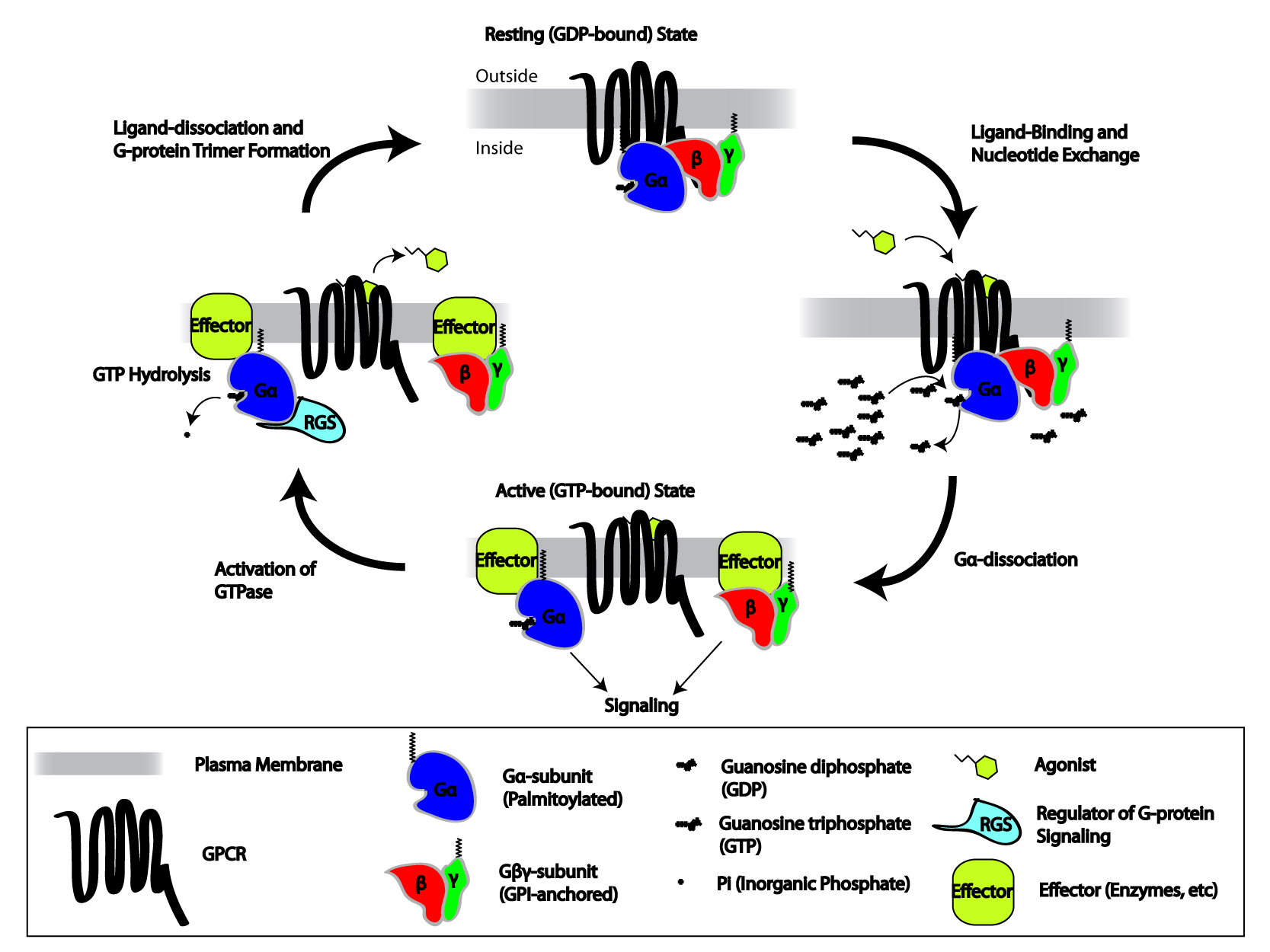
Wikipedia contributors. (2026b, March 23).
G protein-coupled receptor. Retrieved from
https://en.wikipedia.org/wiki/G_protein-coupled_receptor
Wikipedia contributors. (2026c, March 26).
Hodgkin–Huxley model. Retrieved from
https://en.wikipedia.org/wiki/Hodgkin%E2%80%93Huxley_model
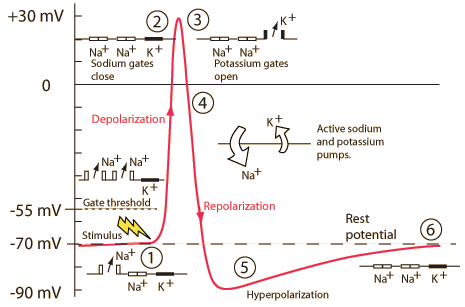
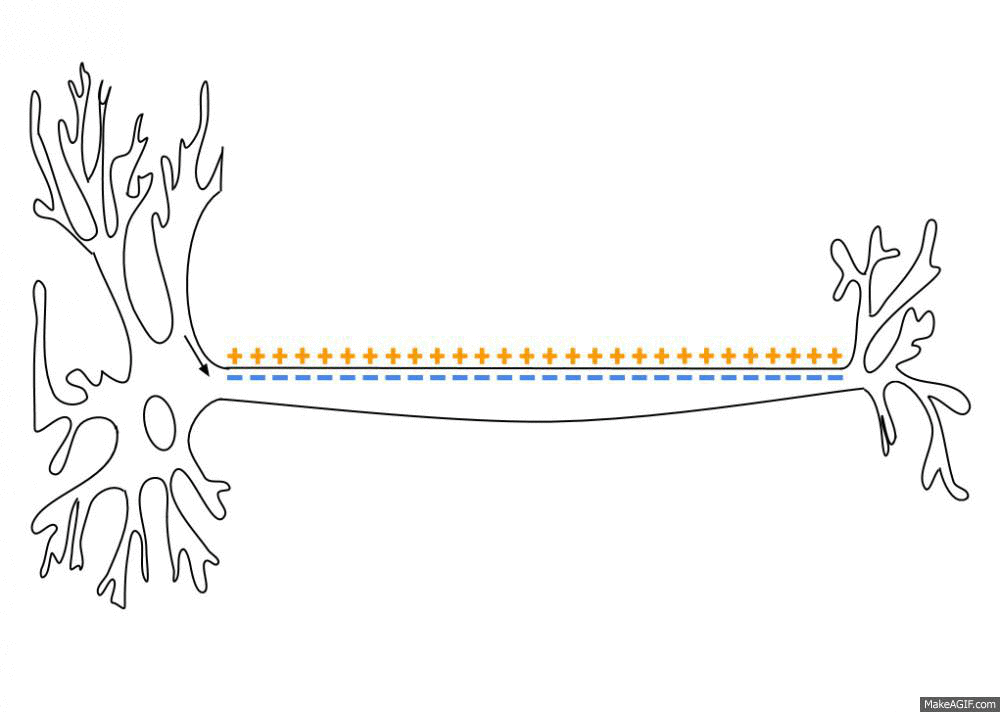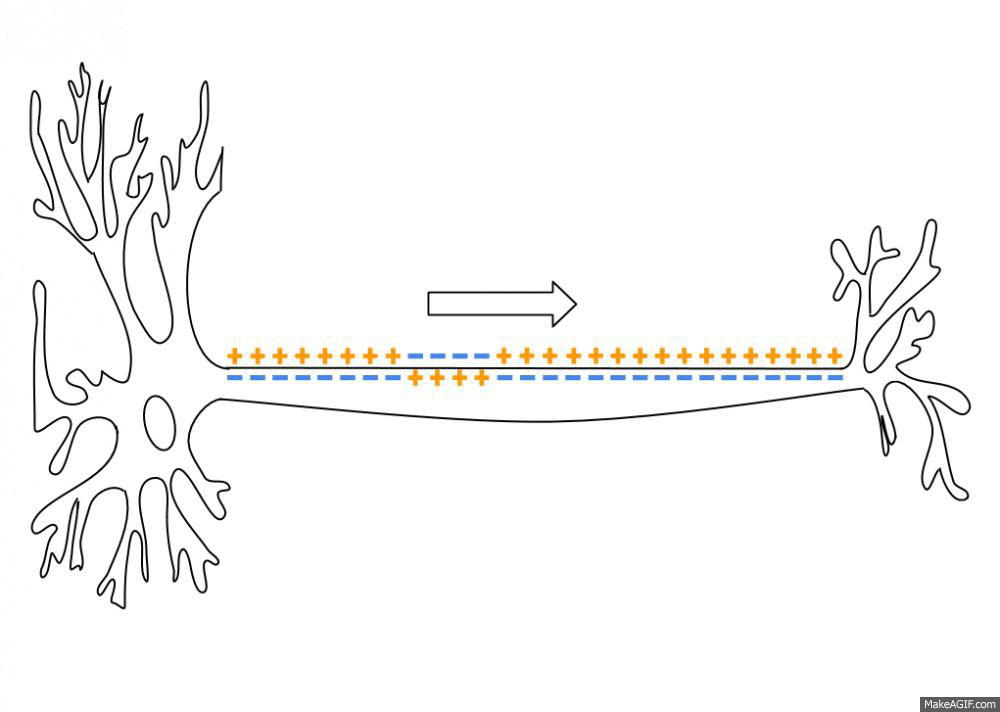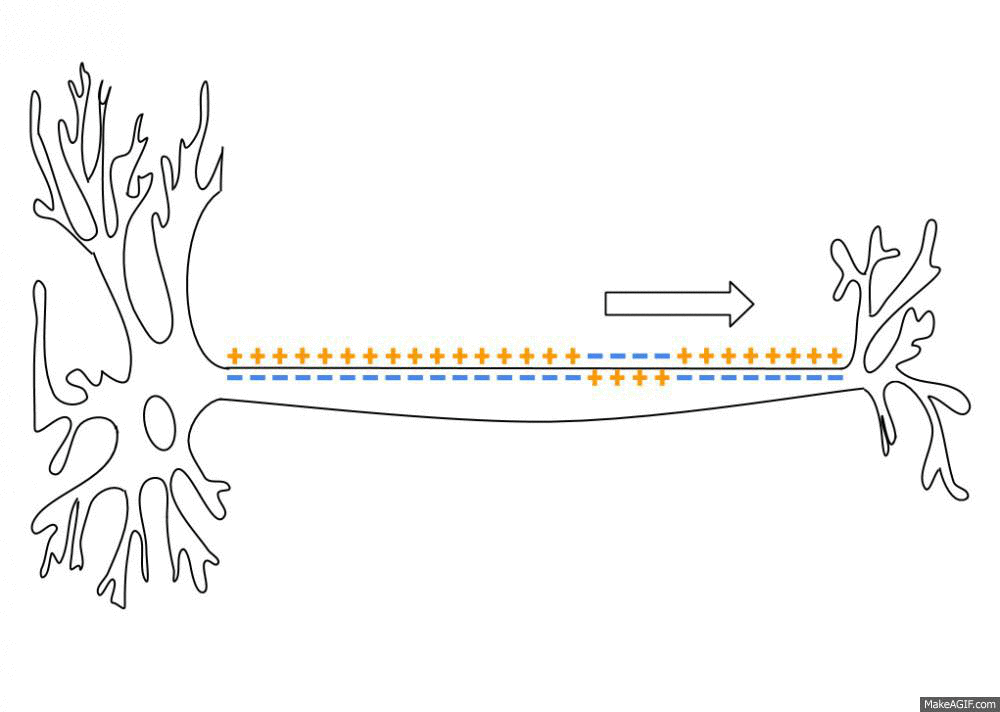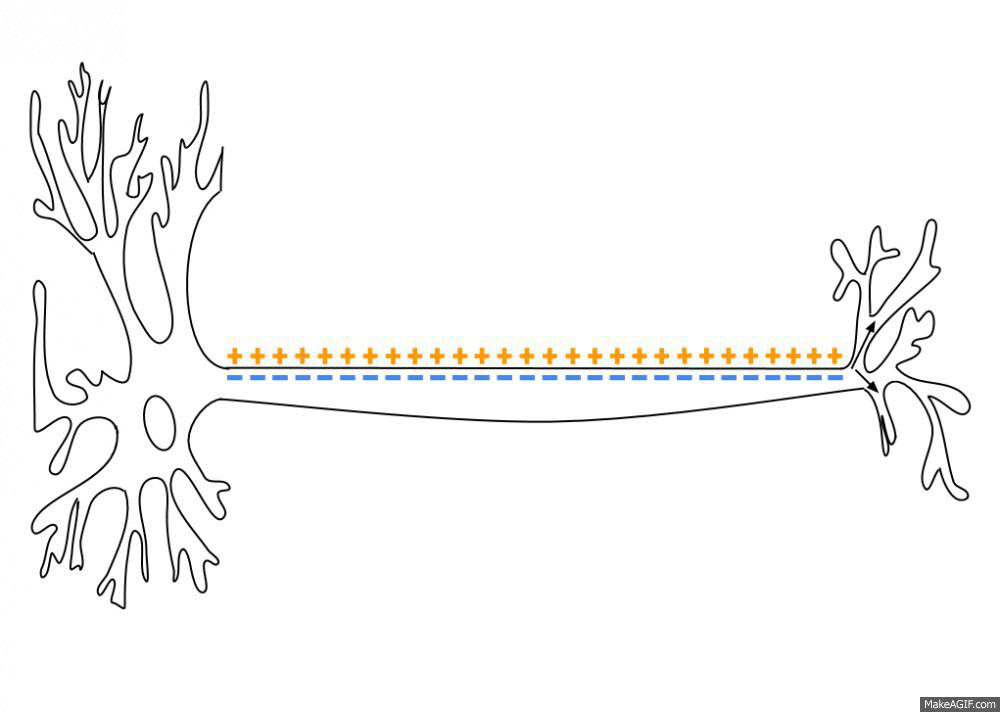
Wikipedia contributors. (2026d, March 30). Action potential. Retrieved from
https://en.wikipedia.org/wiki/Action_potential