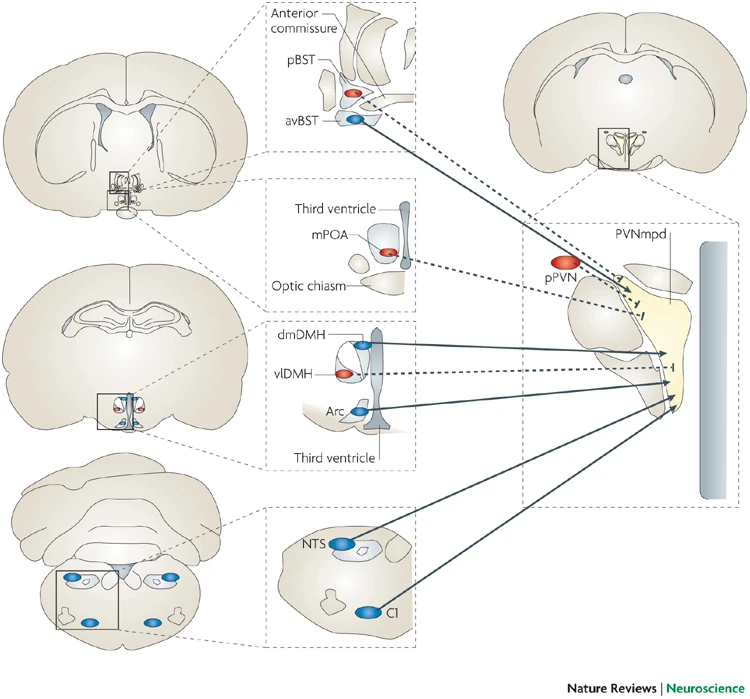
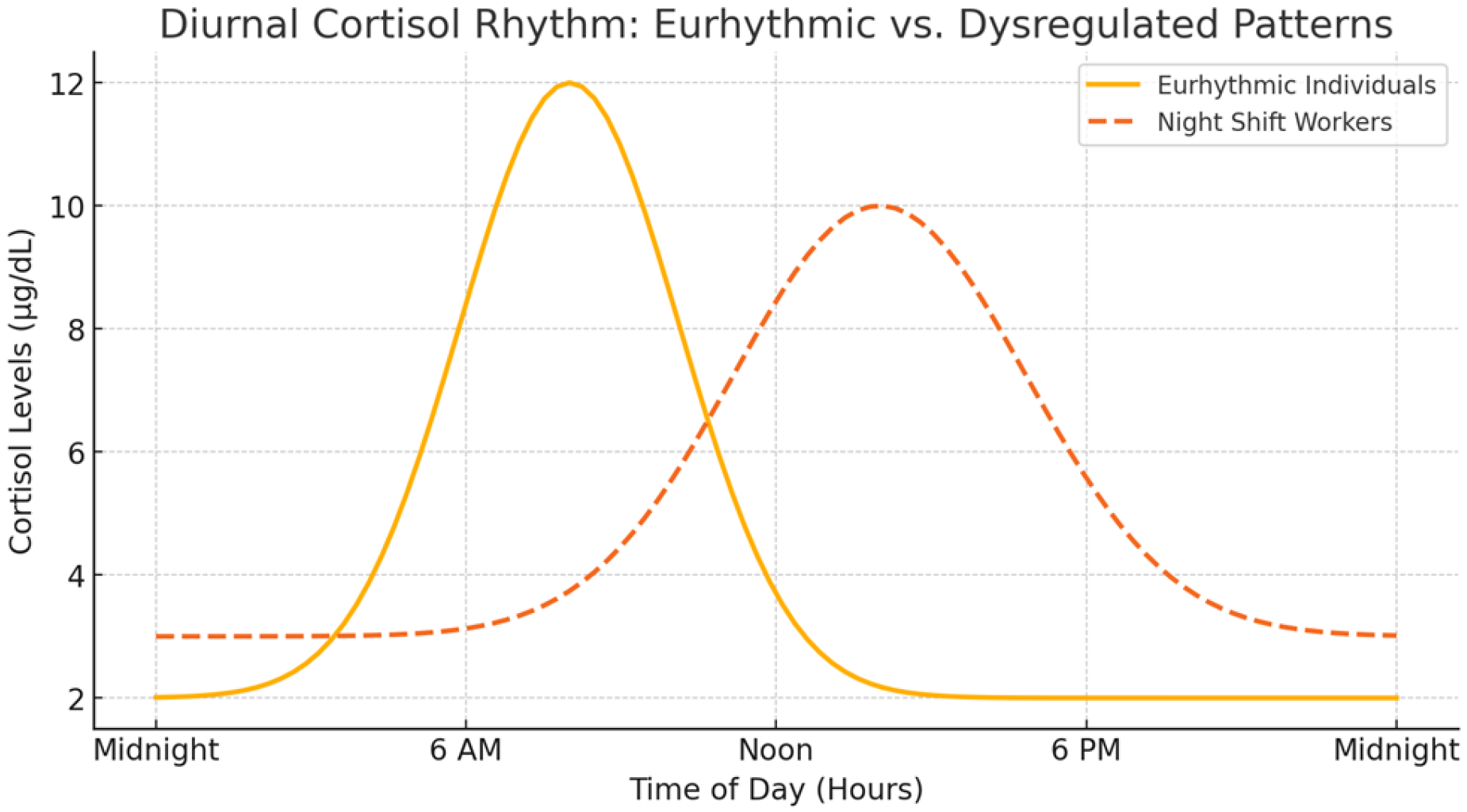
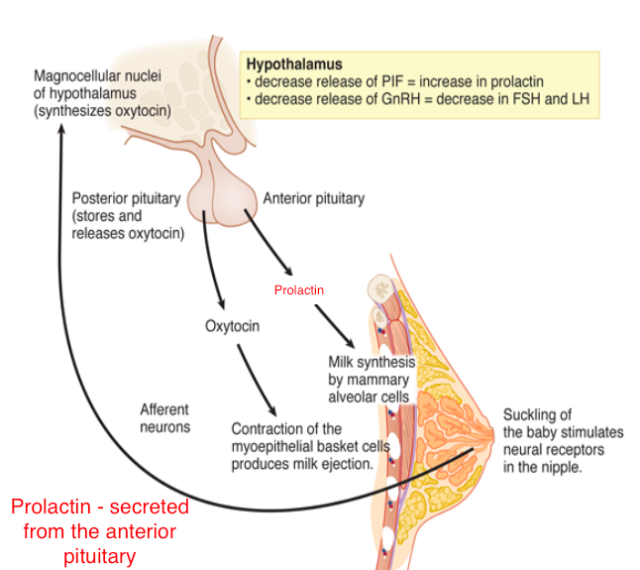
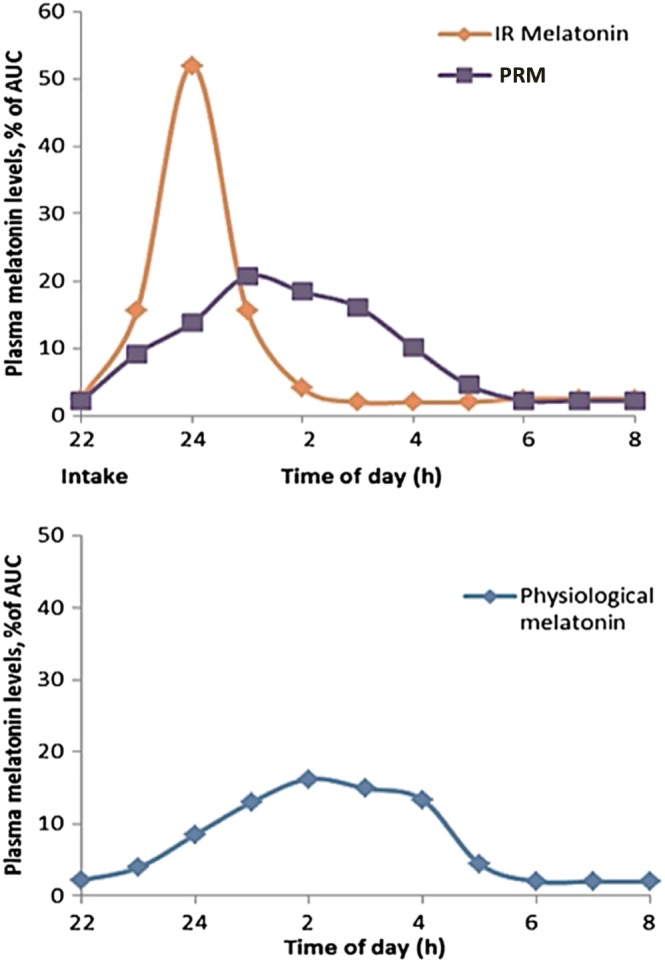
Andreadi, A., Andreadi, S., Todaro, F., Ippoliti, L., Bellia, A., Magrini, A., … Lauro, D. (2025). Modified cortisol circadian rhythm: The hidden toll of night-shift work.
International Journal of Molecular Sciences,
26, 2090.
https://doi.org/10.3390/ijms26052090
Ardura, J., Gutierrez, R., Andres, J., & Agapito, T. (2003). Emergence and evolution of the circadian rhythm of melatonin in children.
Horm. Res.,
59(2), 66–72.
https://doi.org/68571
Arendt, J., & Aulinas, A. (2000). Physiology of the pineal gland and melatonin. In
Endotext. South Dartmouth (MA): MDText.com, Inc. Retrieved from
https://www.ncbi.nlm.nih.gov/books/NBK550972/
Avery, M. C., & Krichmar, J. L. (2017). Neuromodulatory systems and their interactions: A review of models, theories, and experiments.
Frontiers in Neural Circuits,
11, 108.
https://doi.org/10.3389/fncir.2017.00108
Avram, M., Grothe, M. J., Meinhold, L., Leucht, C., Leucht, S., Borgwardt, S., … Sorg, C. (2021). Lower cholinergic basal forebrain volumes link with cognitive difficulties in schizophrenia.
Neuropsychopharmacology: Official Publication of the American College of Neuropsychopharmacology.
https://doi.org/10.1038/s41386-021-01070-x
Carhart-Harris, R. L., & Nutt, D. J. (2017). Serotonin and brain function: A tale of two receptors.
Journal of Psychopharmacology,
31(9), 1091–1120.
https://doi.org/10.1177/0269881117725915
De Ponti, F. (2004). Pharmacology of serotonin: What a clinician should know.
Gut,
53(10), 1520–1535.
https://doi.org/10.1136/gut.2003.035568
Deussing, J. M., & Chen, A. (2018). The
Corticotropin-Releasing factor family: Physiology of the stress response.
Physiological Reviews,
98(4), 2225–2286.
https://doi.org/10.1152/physrev.00042.2017
Domes, G., Heinrichs, M., Kumbier, E., Grossmann, A., Hauenstein, K., & Herpertz, S. C. (2013). Effects of intranasal oxytocin on the neural basis of face processing in autism spectrum disorder.
Biological Psychiatry,
74(3), 164–171. https://doi.org/
http://dx.doi.org/10.1016/j.biopsych.2013.02.007
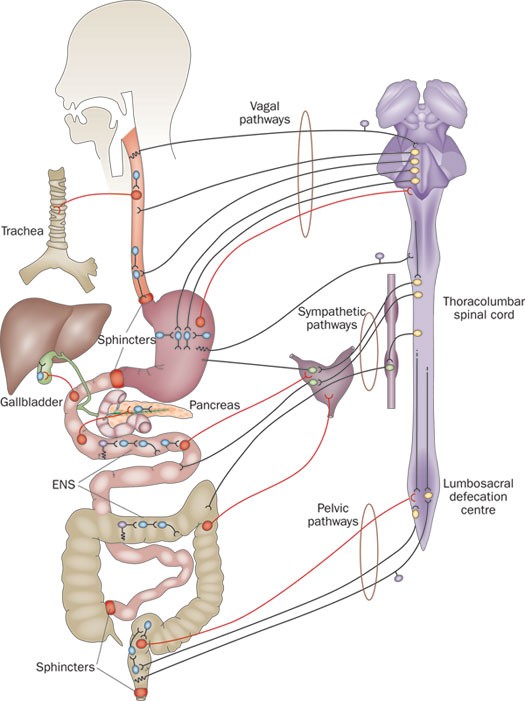
Furness, J. B. (2012). The enteric nervous system and neurogastroenterology.
Nature Reviews. Gastroenterology & Hepatology,
9(5), 286–294.
https://doi.org/10.1038/nrgastro.2012.32
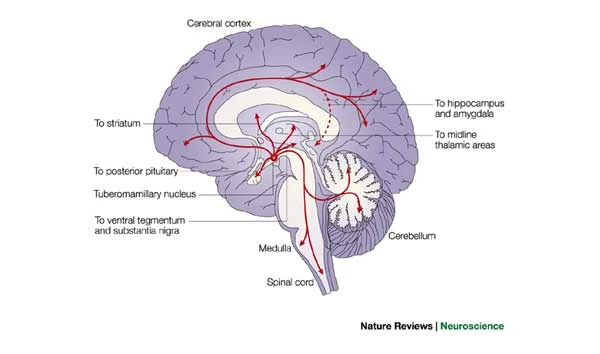
Haas, H., & Panula, P. (2003). The role of histamine and the tuberomamillary nucleus in the nervous system.
Nature Reviews. Neuroscience,
4(2), 121–130.
https://doi.org/10.1038/nrn1034
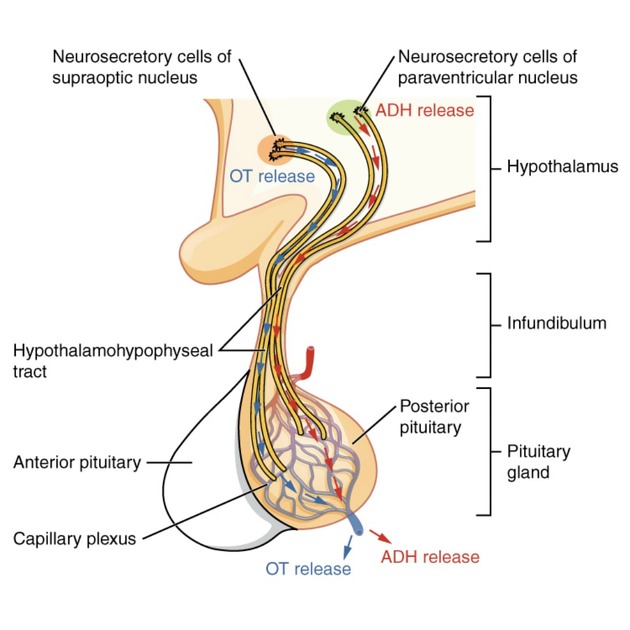
Hacking, C., Gaillard, F., & Smith, D. (2016, December 7). Posterior pituitary. In
Radiopaedia.org. Radiopaedia.org.
https://doi.org/10.53347/rid-49865
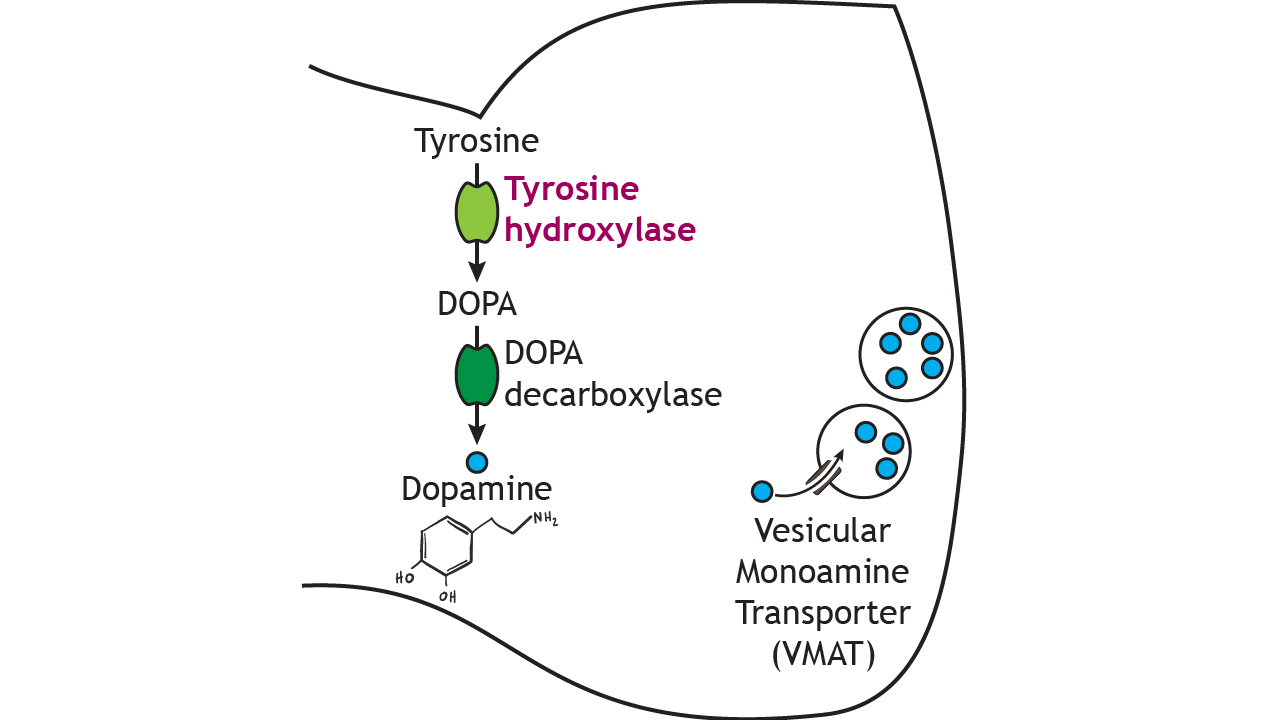
Henley, C. (2021). Neurotransmitter synthesis and storage. In
Foundations of neuroscience. Michigan State University Libraries. Retrieved from
https://openbooks.lib.msu.edu/neuroscience/chapter/neurotransmitter-synthesis-and-storage/
Małgorzata, P., Paweł, K., Iwona, M. L., Brzostek, T., & Andrzej, P. (2020). Glutamatergic dysregulation in mood disorders: Opportunities for the discovery of novel drug targets.
Expert Opinion on Therapeutic Targets,
24(12), 1187–1209.
https://doi.org/10.1080/14728222.2020.1836160
McCutcheon, R. A., Krystal, J. H., & Howes, O. D. (2020). Dopamine and glutamate in schizophrenia: Biology, symptoms and treatment.
World Psychiatry: Official Journal of the World Psychiatric Association,
19(1), 15–33.
https://doi.org/10.1002/wps.20693
Mesulam, M.-M. (2013). Cholinergic circuitry of the human nucleus basalis and its fate in alzheimer’s disease.
The Journal of Comparative Neurology,
521(18), 4124–4144.
https://doi.org/10.1002/cne.23415
Moncrieff, J., Cooper, R. E., Stockmann, T., Amendola, S., Hengartner, M. P., & Horowitz, M. A. (2022). The serotonin theory of depression: A systematic umbrella review of the evidence.
Molecular Psychiatry.
https://doi.org/10.1038/s41380-022-01661-0
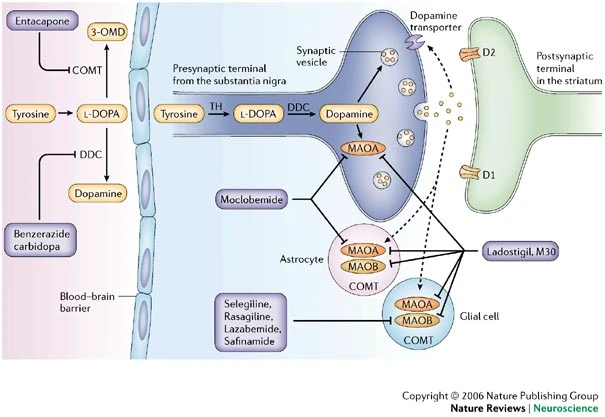
Overk, C. R., & Mufson, E. J. (2010). Dopamine transporter: Aging and parkinson’s disease. In
Encyclopedia of movement disorders (pp. 330–332). Elsevier.
https://doi.org/10.1016/b978-0-12-374105-9.00237-9
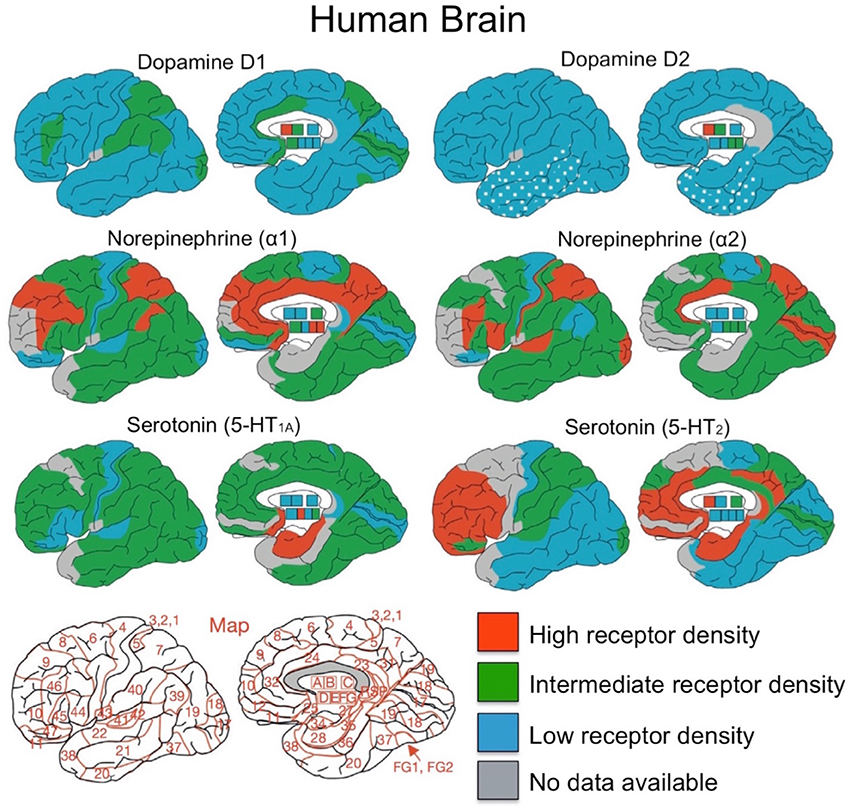
Palomero-Gallagher, N., Amunts, K., & Zilles, K. (2015). Transmitter receptor distribution in the human brain. In
Brain mapping (pp. 261–275). Elsevier.
https://doi.org/10.1016/b978-0-12-397025-1.00221-9
Picciotto, M. R., Higley, M. J., & Mineur, Y. S. (2012). Acetylcholine as a neuromodulator: Cholinergic signaling shapes nervous system function and behavior.
Neuron,
76, 116–129.
https://doi.org/10.1016/j.neuron.2012.08.036
Ren, J., Friedmann, D., Xiong, J., Liu, C. D., Ferguson, B. R., Weerakkody, T., … Luo, L. (2018). Anatomically defined and functionally distinct dorsal raphe serotonin sub-systems.
Cell.
https://doi.org/10.1016/j.cell.2018.07.043
Songs, M. (2018).
Muppet songs: Mahna mahna (muppet show - 1976). You
Tube. Retrieved from
https://www.youtube.com/watch?v=TbZ_hTEOKZc
Ulrich-Lai, Y. M., & Herman, J. P. (2009). Neural regulation of endocrine and autonomic stress responses.
Nature Reviews Neuroscience,
10(6), 397–409.
https://doi.org/10.1038/nrn2647
Viviani, D., Charlet, A., Burg, E. van den, Robinet, C., Hurni, N., Abatis, M., … Stoop, R. (2011). Oxytocin selectively gates fear responses through distinct outputs from the central amygdala.
Science,
333(6038), 104–107.
https://doi.org/10.1126/science.1201043
Vogelsang, D. A., & D’Esposito, M. (2018). Is there evidence for a rostral-caudal gradient in fronto-striatal loops and what role does dopamine play?
Frontiers in Neuroscience,
12, 242.
https://doi.org/10.3389/fnins.2018.00242
Weisman, O., & Feldman, R. (2013). Oxytocin effects on the human brain: Findings, questions, and future directions.
Biological Psychiatry,
74(3), 158–159. https://doi.org/
http://dx.doi.org/10.1016/j.biopsych.2013.05.026
What is a neurotransmitter? (n.d.). Retrieved April 8, 2026, from
https://nigms.nih.gov/biobeat/2024/08/what-is-a-neurotransmitter
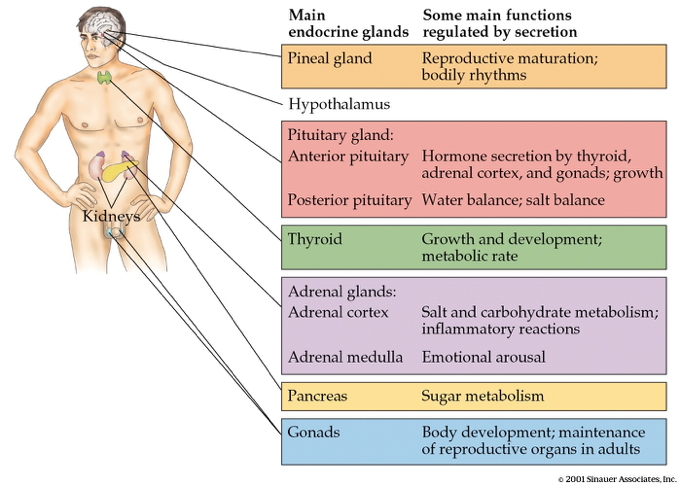
Wikipedia contributors. (2025a, September 28). Adrenal gland. Retrieved from
https://en.wikipedia.org/wiki/Adrenal_gland
Wikipedia contributors. (2025b, October 25). Blind men and an elephant. Retrieved from
https://en.wikipedia.org/wiki/Blind_men_and_an_elephant
Wikipedia contributors. (2025c, November 20). Cycloplegia. Retrieved from
https://en.wikipedia.org/wiki/Cycloplegia#Cycloplegic_medication
Wikipedia contributors. (2025d, November 24). Raphe nuclei. Retrieved from
https://en.wikipedia.org/wiki/Raphe_nuclei
Wikipedia contributors. (2026a, February 23). Monoamine oxidase inhibitor. Retrieved from
https://en.wikipedia.org/wiki/Monoamine_oxidase_inhibitor
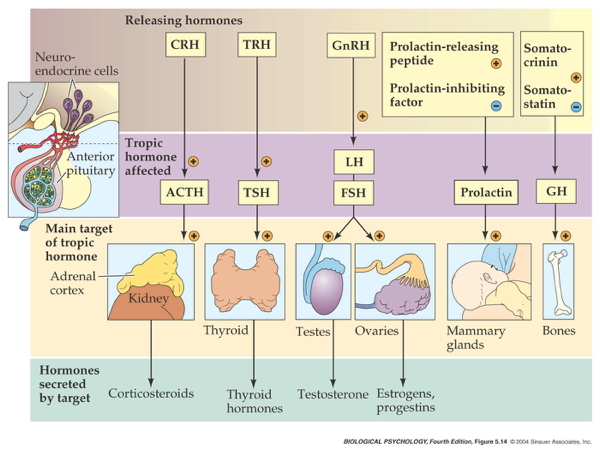
Wikipedia contributors. (2026b, March 26). Anterior pituitary. Retrieved from
https://en.wikipedia.org/wiki/Anterior_pituitary
Youdim, M. B. H., Edmondson, D., & Tipton, K. F. (2006). The therapeutic potential of monoamine oxidase inhibitors.
Nature Reviews. Neuroscience,
7(4), 295–309.
https://doi.org/10.1038/nrn1883
Zisapel, N. (2018). New perspectives on the role of melatonin in human sleep, circadian rhythms and their regulation: Melatonin in human sleep and circadian rhythms.
British Journal of Pharmacology,
175, 3190–3199.
https://doi.org/10.1111/bph.14116
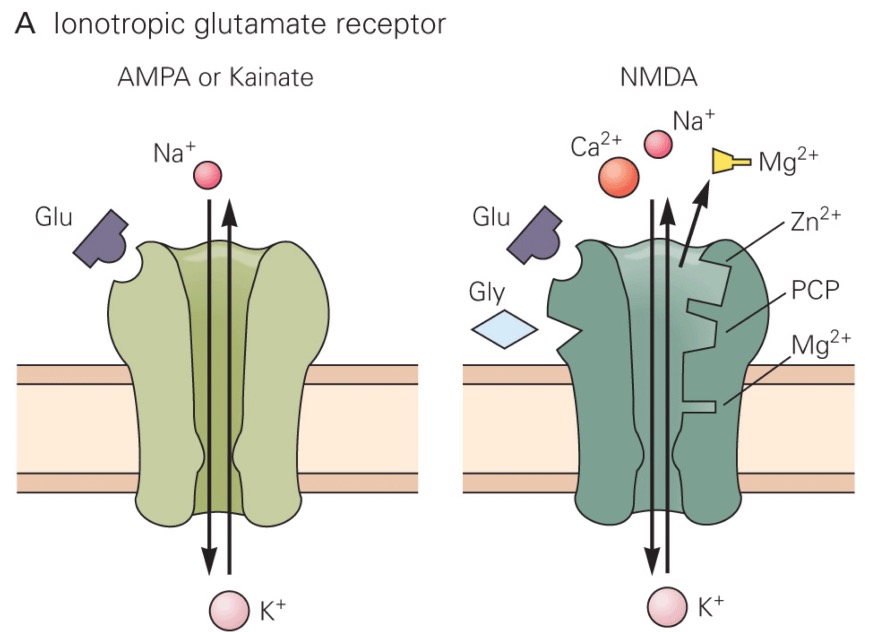

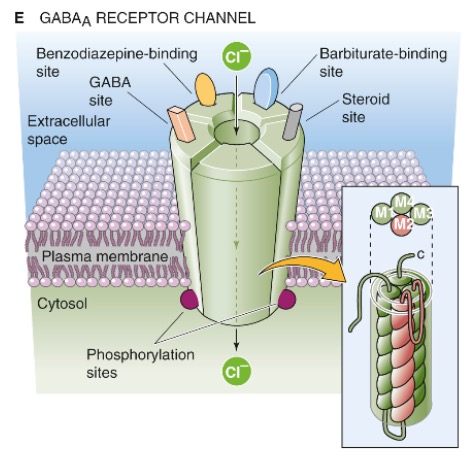
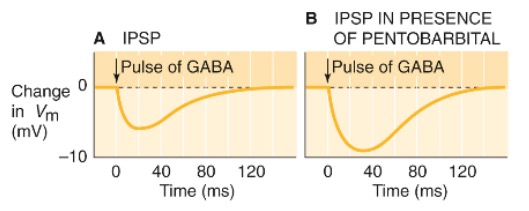

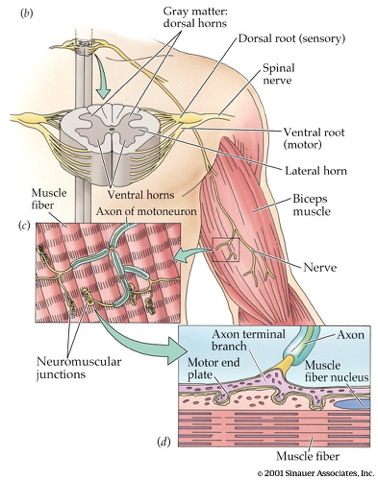
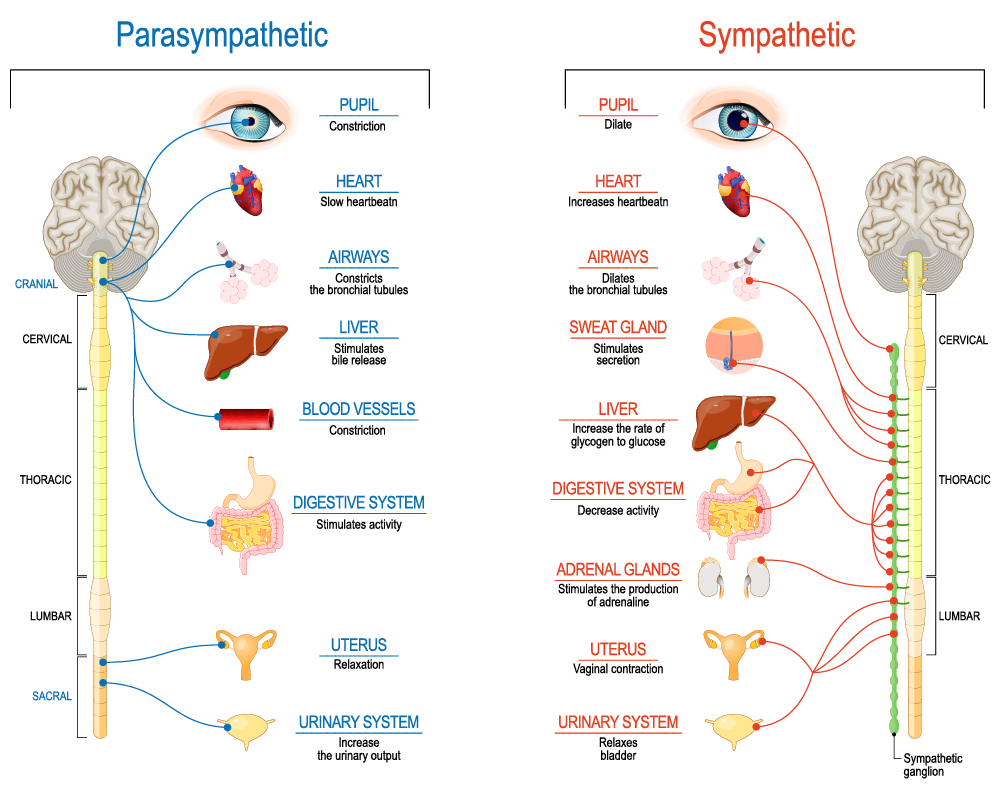



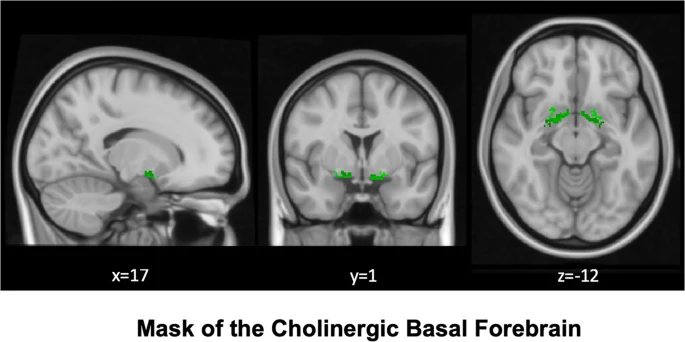
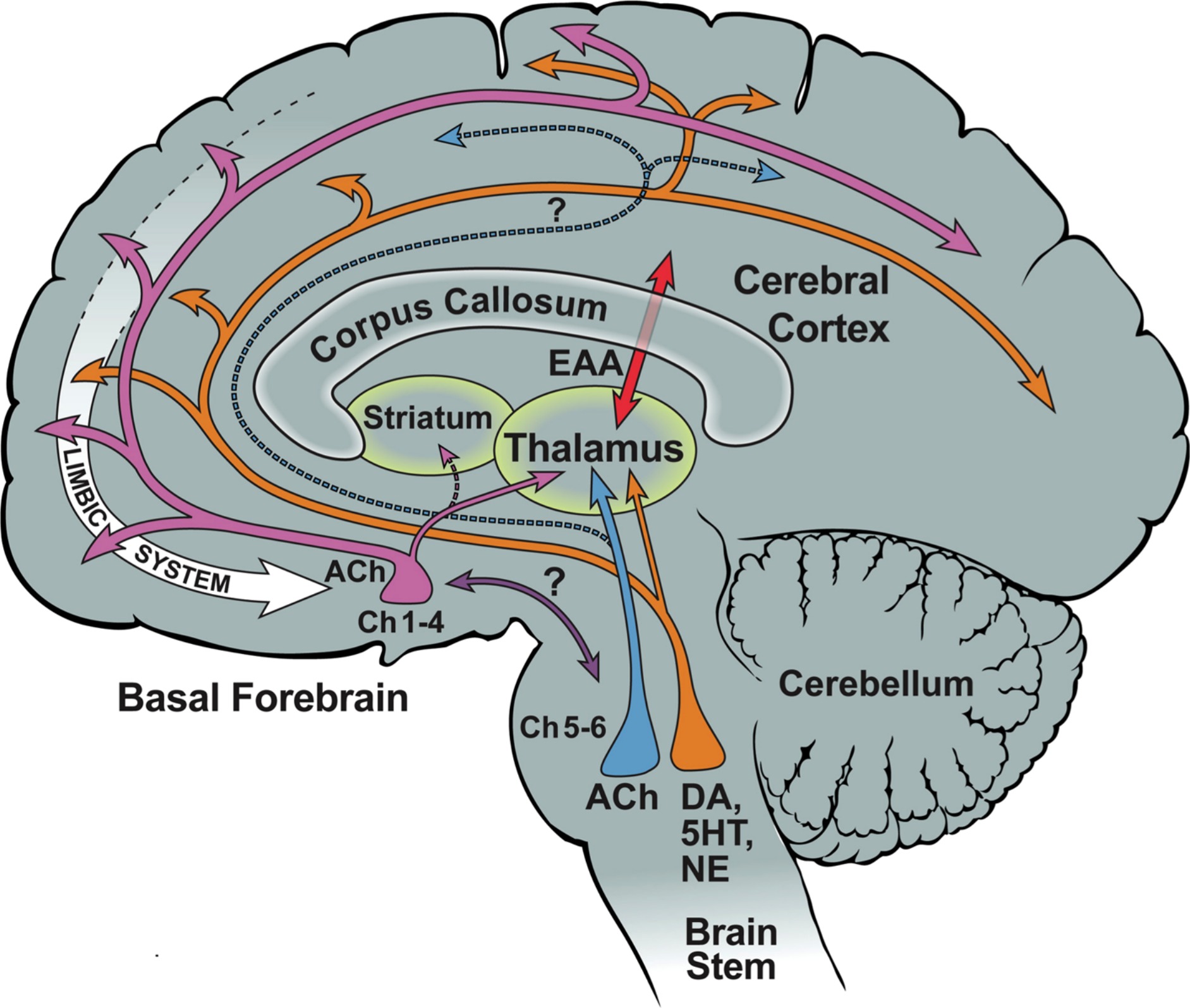
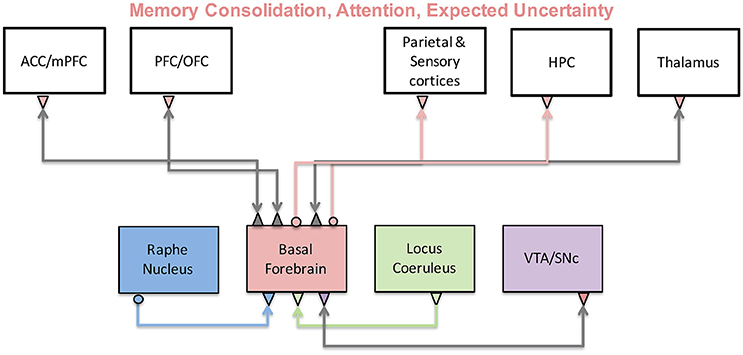














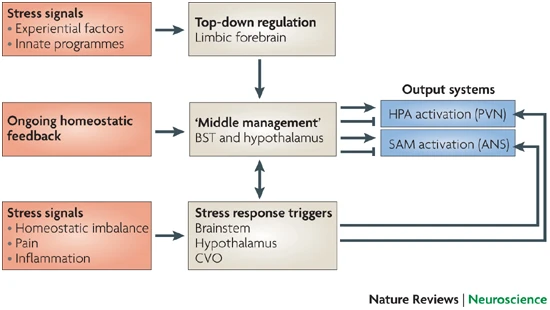
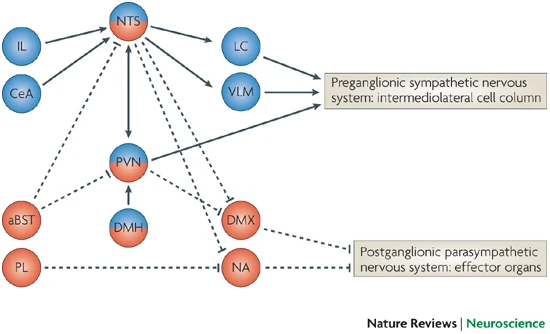 }
}