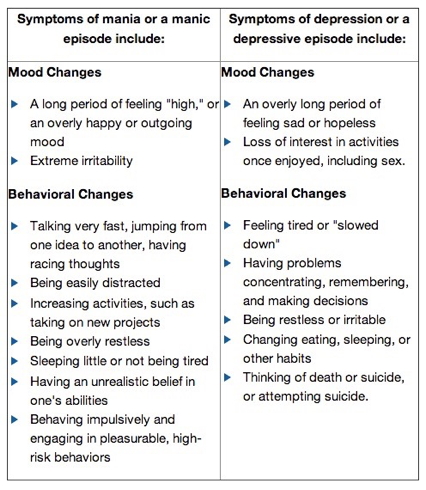
Baldessarini, R. J., Pompili, M., & Tondo, L. (2006). Suicide in
Bipolar Disorder:
Risks and
Management.
CNS Spectrums,
11(06), 465–471.
https://doi.org/10.1017/S1092852900014681
Bi, B., Che, D., & Bai, Y. (2022). Neural network of bipolar disorder: Toward integration of neuroimaging and neurocircuit-based treatment strategies.
Translational Psychiatry,
12(1), 143.
https://doi.org/10.1038/s41398-022-01917-x
Bisso, E., Signorelli, M. S., Milazzo, M., Maglia, M., Polosa, R., Aguglia, E., & Caponnetto, P. (2020). Immersive virtual reality applications in schizophrenia spectrum therapy: A systematic review.
International Journal of Environmental Research and Public Health,
17(17).
https://doi.org/10.3390/ijerph17176111
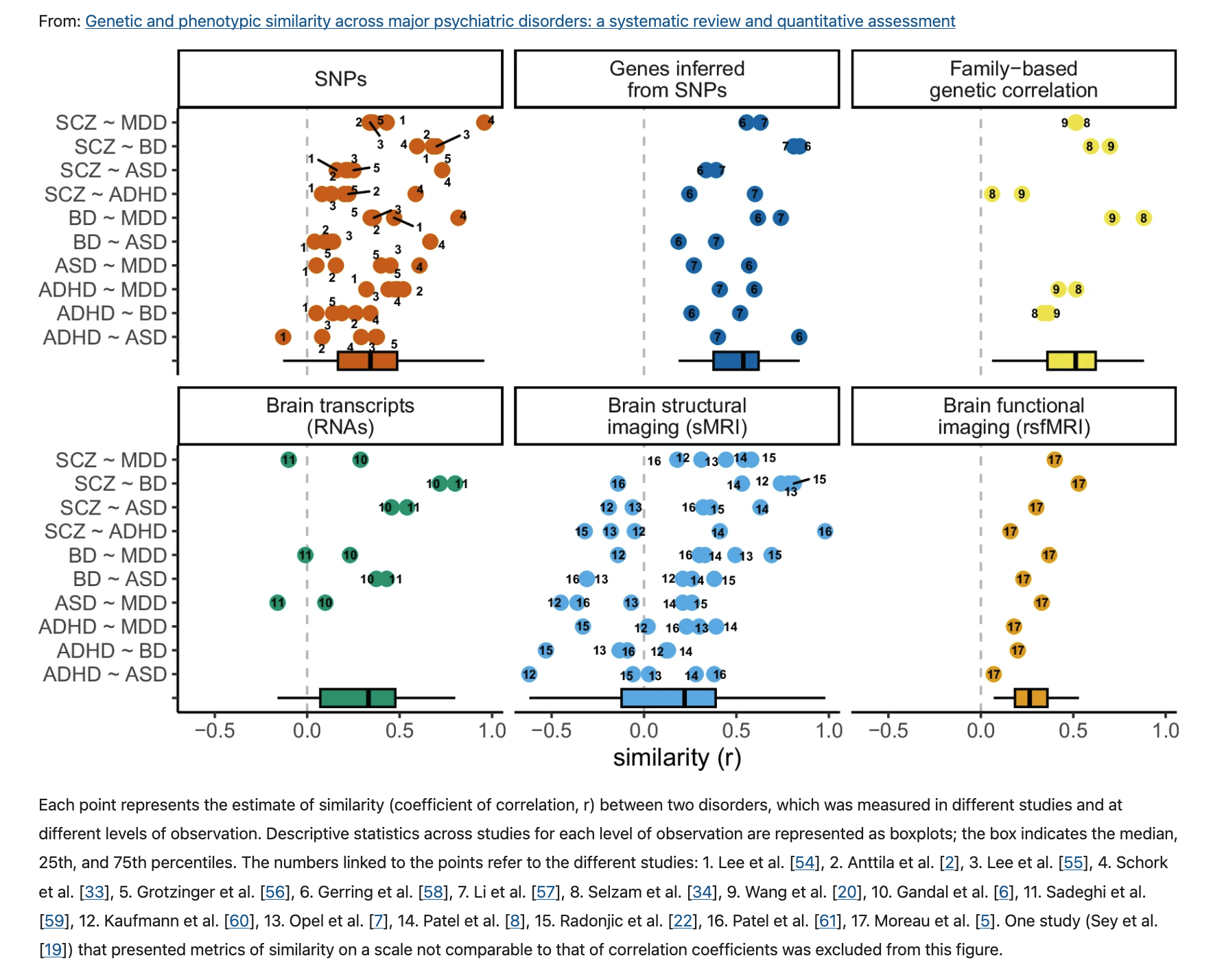
Bourque, V.-R., Poulain, C., Proulx, C., Moreau, C. A., Joober, R., Forgeot d’Arc, B., … Jacquemont, S. (2024). Genetic and phenotypic similarity across major psychiatric disorders: A systematic review and quantitative assessment.
Translational Psychiatry,
14, 171.
https://doi.org/10.1038/s41398-024-02866-3
Brown, A. S., & Derkits, E. J. (2010). Prenatal infection and schizophrenia: A review of epidemiologic and translational studies.
The American Journal of Psychiatry,
167, 261–280.
https://doi.org/10.1176/appi.ajp.2009.09030361
Cantor-Graae, E., & Selten, J.-P. (2005). Schizophrenia and migration: A meta-analysis and review.
The American Journal of Psychiatry,
162(1), 12–24.
https://doi.org/10.1176/appi.ajp.162.1.12
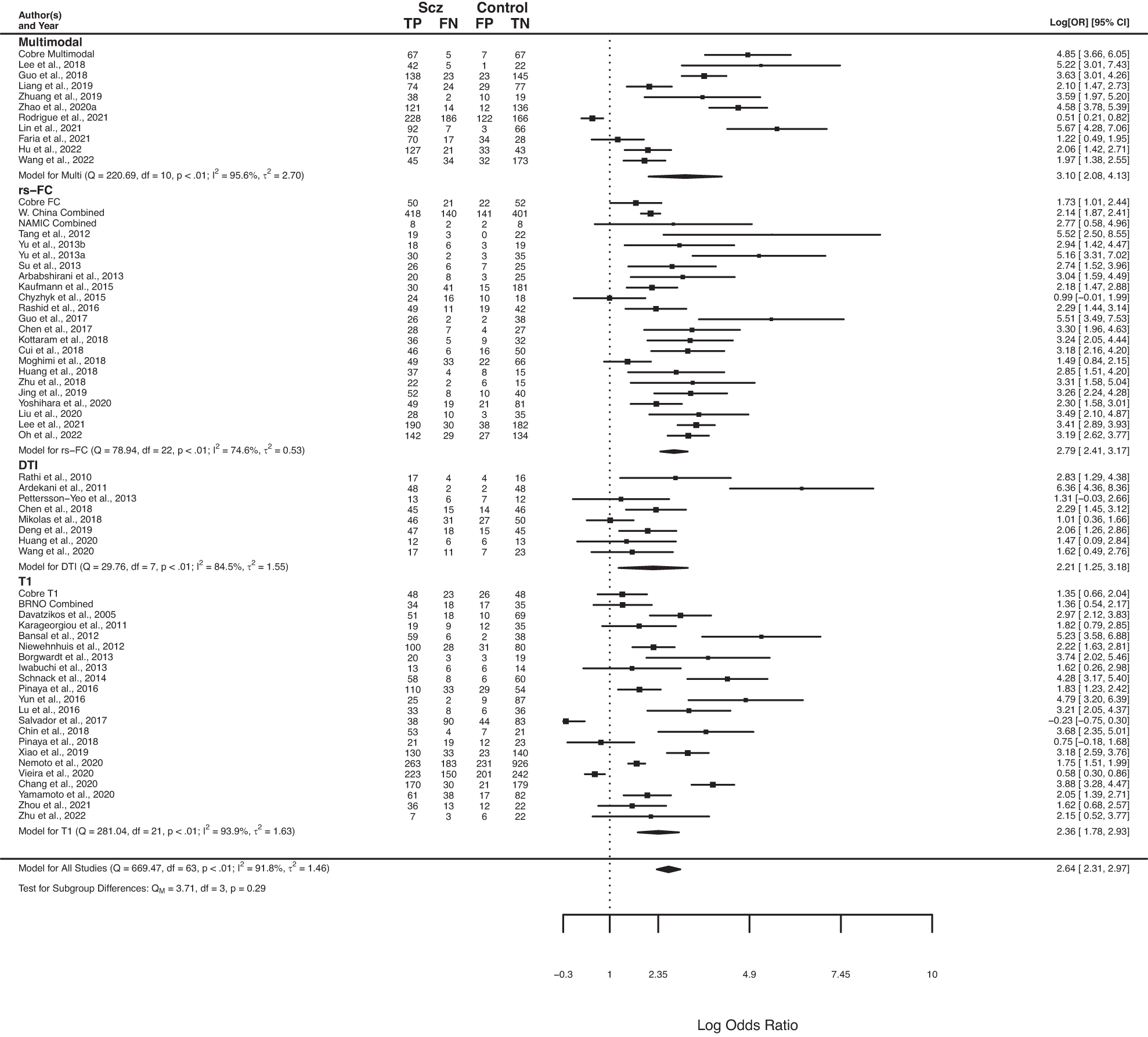
Ching, C. R. K., Hibar, D. P., Gurholt, T. P., Nunes, A., Thomopoulos, S. I., Abé, C., … ENIGMA Bipolar Disorder Working Group. (2022). What we learn about bipolar disorder from large-scale neuroimaging: Findings and future directions from the
ENIGMA bipolar disorder working group.
Human Brain Mapping,
43(1), 56–82.
https://doi.org/10.1002/hbm.25098
Craddock, N., & Sklar, P. (2013). Genetics of bipolar disorder.
The Lancet,
381(9878), 1654–1662.
https://doi.org/10.1016/S0140-6736(13)60855-7
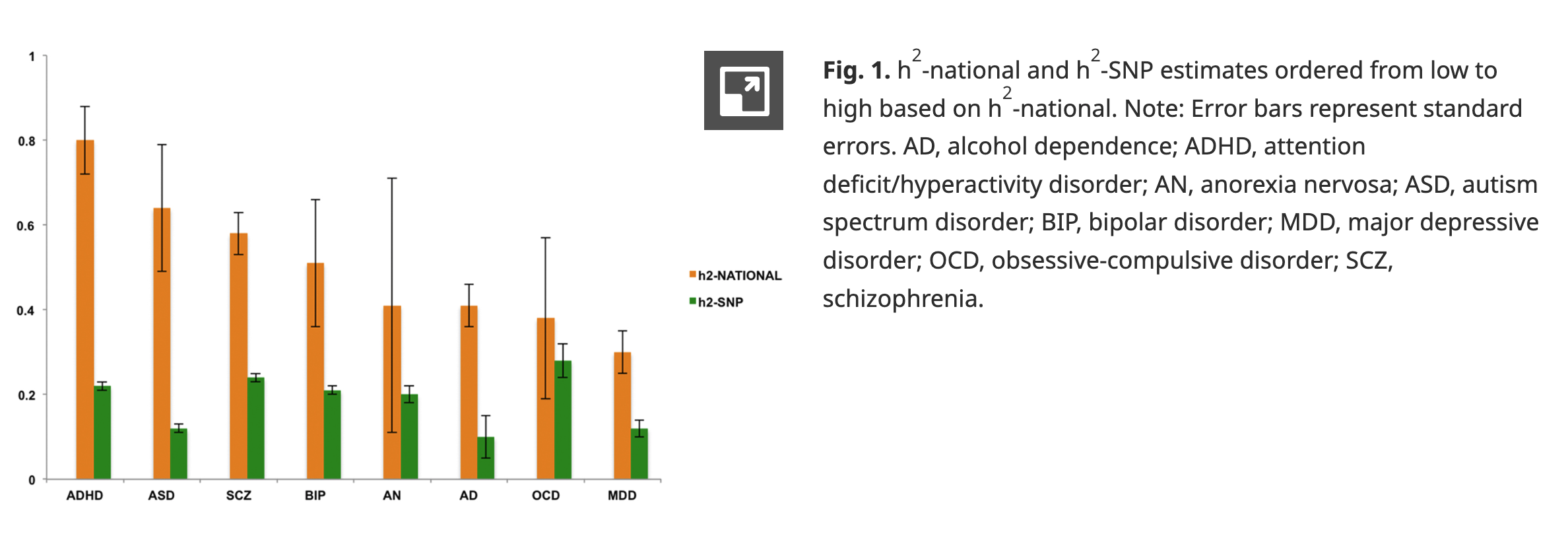
Cross-Disorder Group of the Psychiatric Genomics Consortium. (2013). Identification of risk loci with shared effects on five major psychiatric disorders: A genome-wide analysis.
The Lancet,
381(9875), 1371–1379.
https://doi.org/10.1016/S0140-6736(12)62129-1
Davis, K. L., Buchsbaum, M. S., Shihabuddin, L., Spiegel-Cohen, J., Metzger, M., Frecska, E., … Powchik, P. (1998). Ventricular enlargement in poor-outcome schizophrenia.
Biological Psychiatry,
43(11), 783–793.
https://doi.org/10.1016/s0006-3223(97)00553-2
Debost, J.-C., Petersen, L., Grove, J., Hedemand, A., Khashan, A., Henriksen, T., … Mortensen, P. B. (2015). Investigating interactions between early life stress and two single nucleotide polymorphisms in
HSD11B2 on the risk of schizophrenia.
Psychoneuroendocrinology,
60, 18–27.
https://doi.org/10.1016/j.psyneuen.2015.05.013
Dellazizzo, L., Potvin, S., Phraxayavong, K., & Dumais, A. (2021). One-year randomized trial comparing virtual reality-assisted therapy to cognitive-behavioral therapy for patients with treatment-resistant schizophrenia.
NPJ Schizophrenia,
7(1), 9.
https://doi.org/10.1038/s41537-021-00139-2
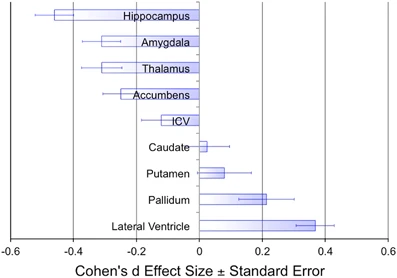
Erp, T. G. M. van, Hibar, D. P., Rasmussen, J. M., Glahn, D. C., Pearlson, G. D., Andreassen, O. A., … Turner, J. A. (2015). Subcortical brain volume abnormalities in 2028 individuals with schizophrenia and 2540 healthy controls via the
ENIGMA consortium.
Mol. Psychiatry.
https://doi.org/10.1038/mp.2015.63
Fornito, A., & Bullmore, E. T. (2015). Reconciling abnormalities of brain network structure and function in schizophrenia.
Curr. Opin. Neurobiol.,
30, 44–50.
https://doi.org/10.1016/j.conb.2014.08.006
Forte, M. F., Oliva, V., De Prisco, M., Garriga, M., Bitanihirwe, B., Alameda, L., … Garcia-Rizo, C. (2024). Obstetric complications and psychopathology in schizophrenia: A systematic review and meta-analysis.
Neuroscience and Biobehavioral Reviews,
167, 105913.
https://doi.org/10.1016/j.neubiorev.2024.105913
Geddes, J. R., & Miklowitz, D. J. (2013). Treatment of bipolar disorder.
The Lancet,
381(9878), 1672–1682.
https://doi.org/10.1016/S0140-6736(13)60857-0
Gitlin, M. J. (2018). Antidepressants in bipolar depression: An enduring controversy.
International Journal of Bipolar Disorders,
6(1), 25.
https://doi.org/10.1186/s40345-018-0133-9
Howes, O. D., Bukala, B. R., & Beck, K. (2023). Schizophrenia: From neurochemistry to circuits, symptoms and treatments.
Nature Reviews. Neurology.
https://doi.org/10.1038/s41582-023-00904-0
Johnson, E. C., Border, R., Melroy-Greif, W. E., Leeuw, C. A. de, Ehringer, M. A., & Keller, M. C. (2017). No evidence that schizophrenia candidate genes are more associated with schizophrenia than noncandidate genes.
Biol. Psychiatry,
82(10), 702–708.
https://doi.org/10.1016/j.biopsych.2017.06.033
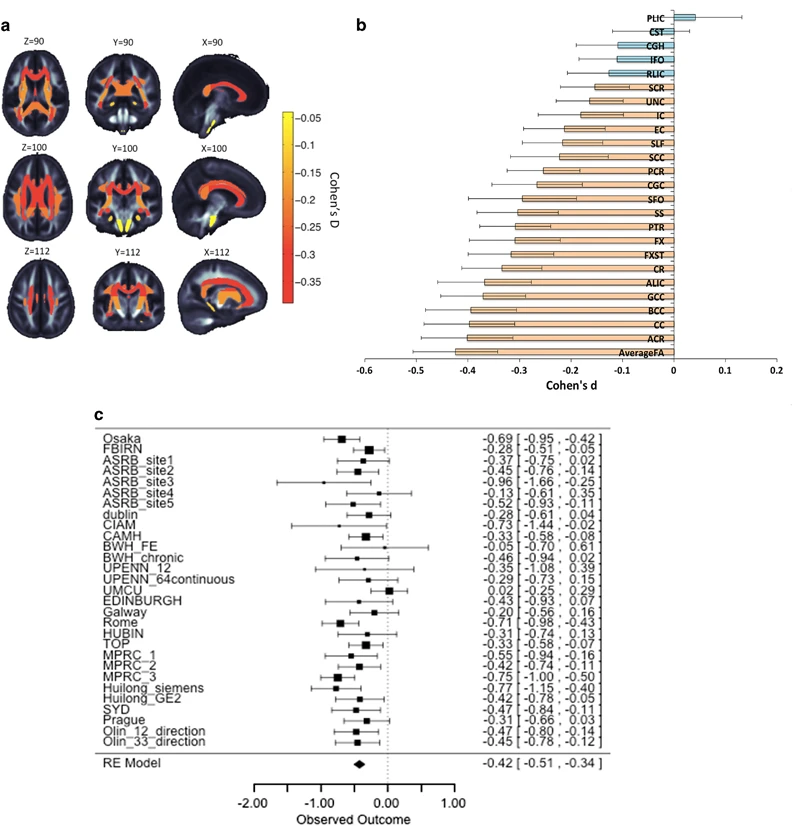
Kelly, S., Jahanshad, N., Zalesky, A., Kochunov, P., Agartz, I., Alloza, C., … Donohoe, G. (2017). Widespread white matter microstructural differences in schizophrenia across 4322 individuals: Results from the
ENIGMA schizophrenia
DTI working group.
Mol. Psychiatry.
https://doi.org/10.1038/mp.2017.170
Kempton, M. J., Stahl, D., Williams, S. C. R., & DeLisi, L. E. (2010). Progressive lateral ventricular enlargement in schizophrenia: A meta-analysis of longitudinal
MRI studies.
Schizophr. Res.,
120(1-3), 54–62.
https://doi.org/10.1016/j.schres.2010.03.036
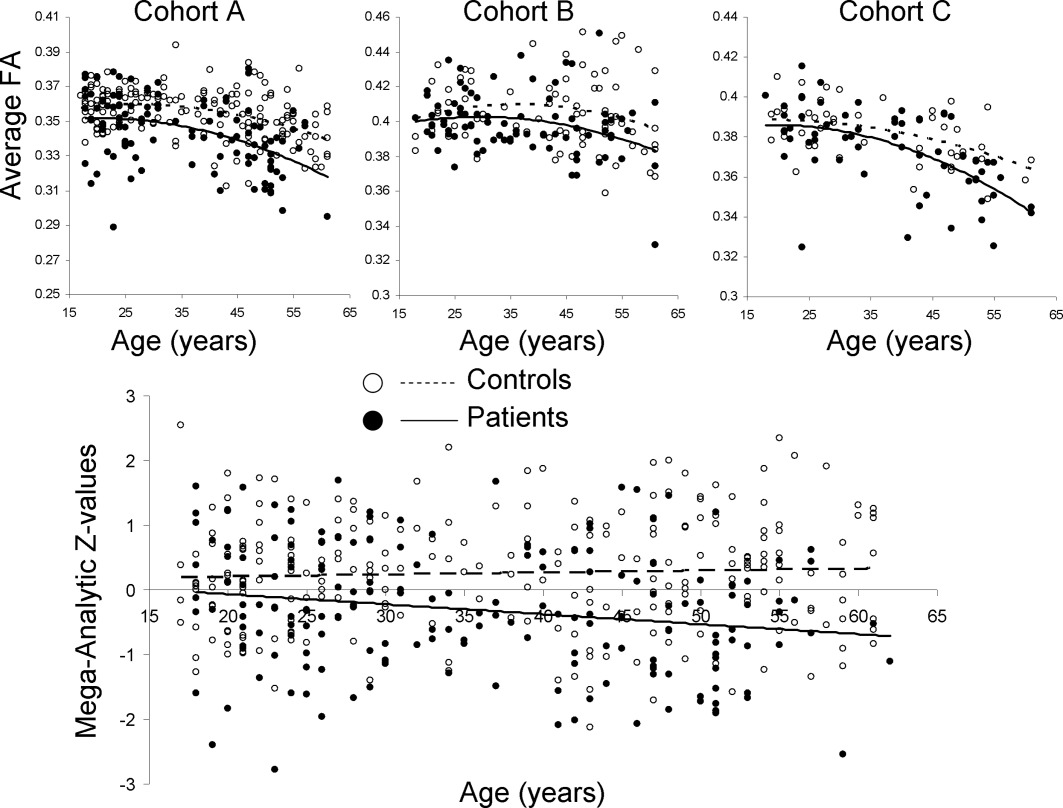
Kochunov, P., Ganjgahi, H., Winkler, A., Kelly, S., Shukla, D. K., Du, X., … Hong, L. E. (2016). Heterochronicity of white matter development and aging explains regional patient control differences in schizophrenia.
Hum. Brain Mapp.,
37(12), 4673–4688.
https://doi.org/10.1002/hbm.23336
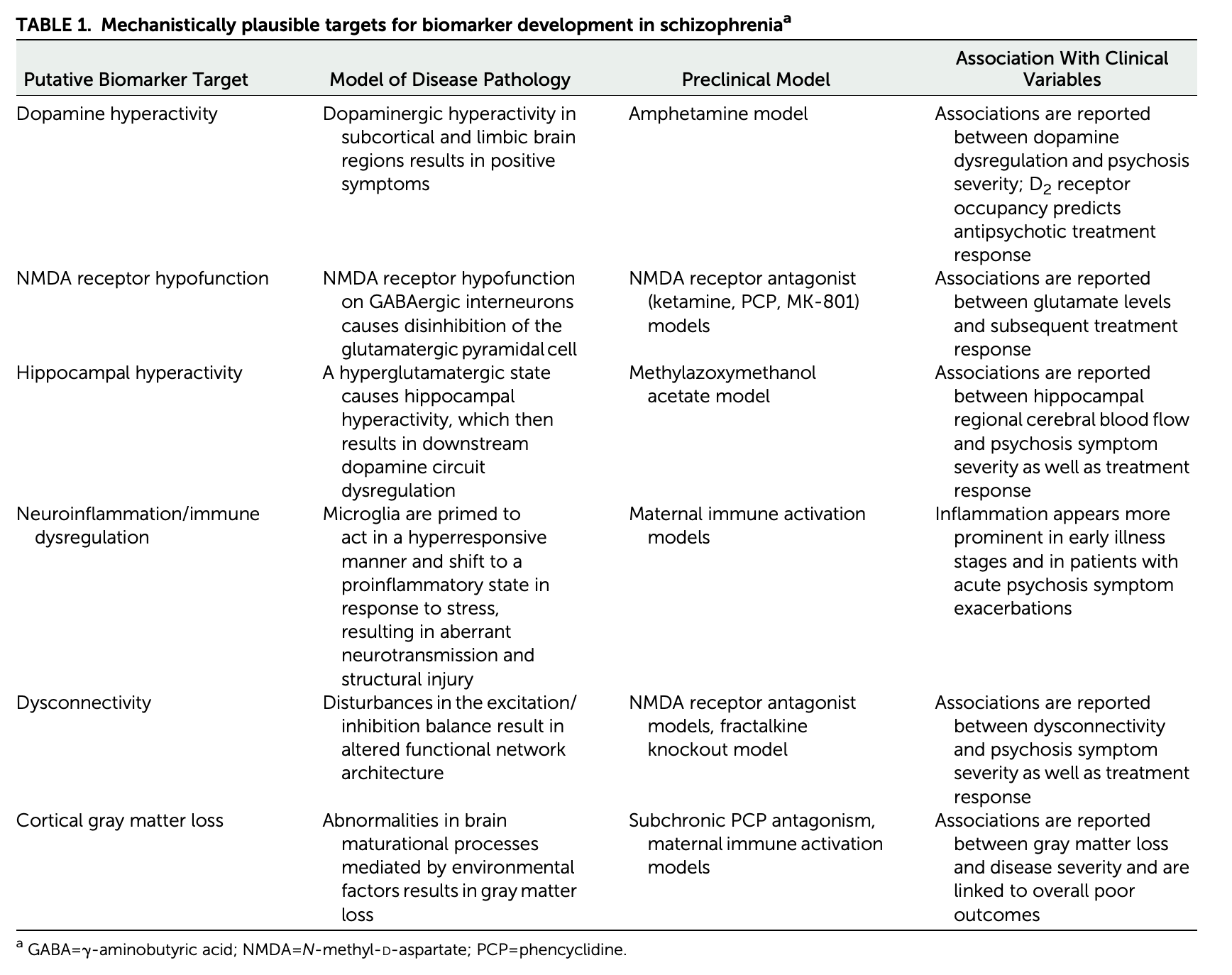
Kraguljac, N. V., McDonald, W. M., Widge, A. S., Rodriguez, C. I., Tohen, M., & Nemeroff, C. B. (2021). Neuroimaging biomarkers in schizophrenia.
The American Journal of Psychiatry,
178(6), 509–521.
https://doi.org/10.1176/appi.ajp.2020.20030340
Levine, S. Z., Levav, I., Pugachova, I., Yoffe, R., & Becher, Y. (2016). Transgenerational effects of genocide exposure on the risk and course of schizophrenia:
A population-based study.
Schizophrenia Research,
176(2), 540–545.
https://doi.org/10.1016/j.schres.2016.06.019
Lv, J., Di Biase, M., Cash, R. F. H., Cocchi, L., Cropley, V. L., Klauser, P., … Zalesky, A. (2021). Individual deviations from normative models of brain structure in a large cross-sectional schizophrenia cohort.
Molecular Psychiatry,
26(7), 3512–3523.
https://doi.org/10.1038/s41380-020-00882-5
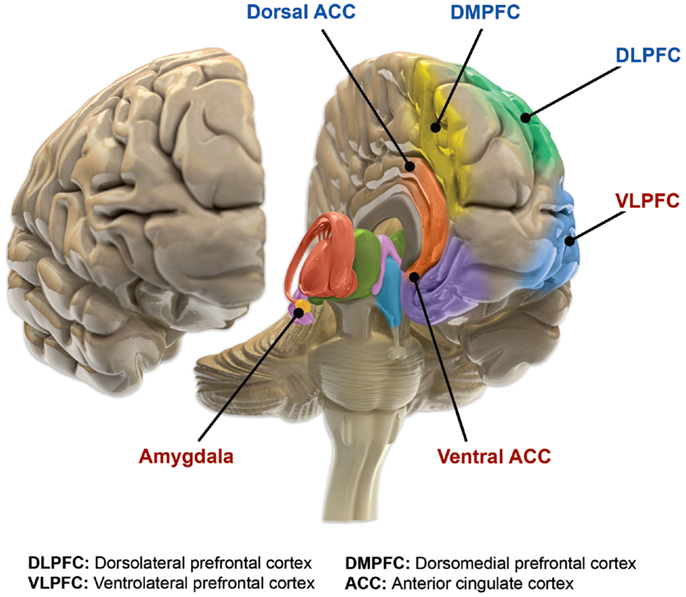
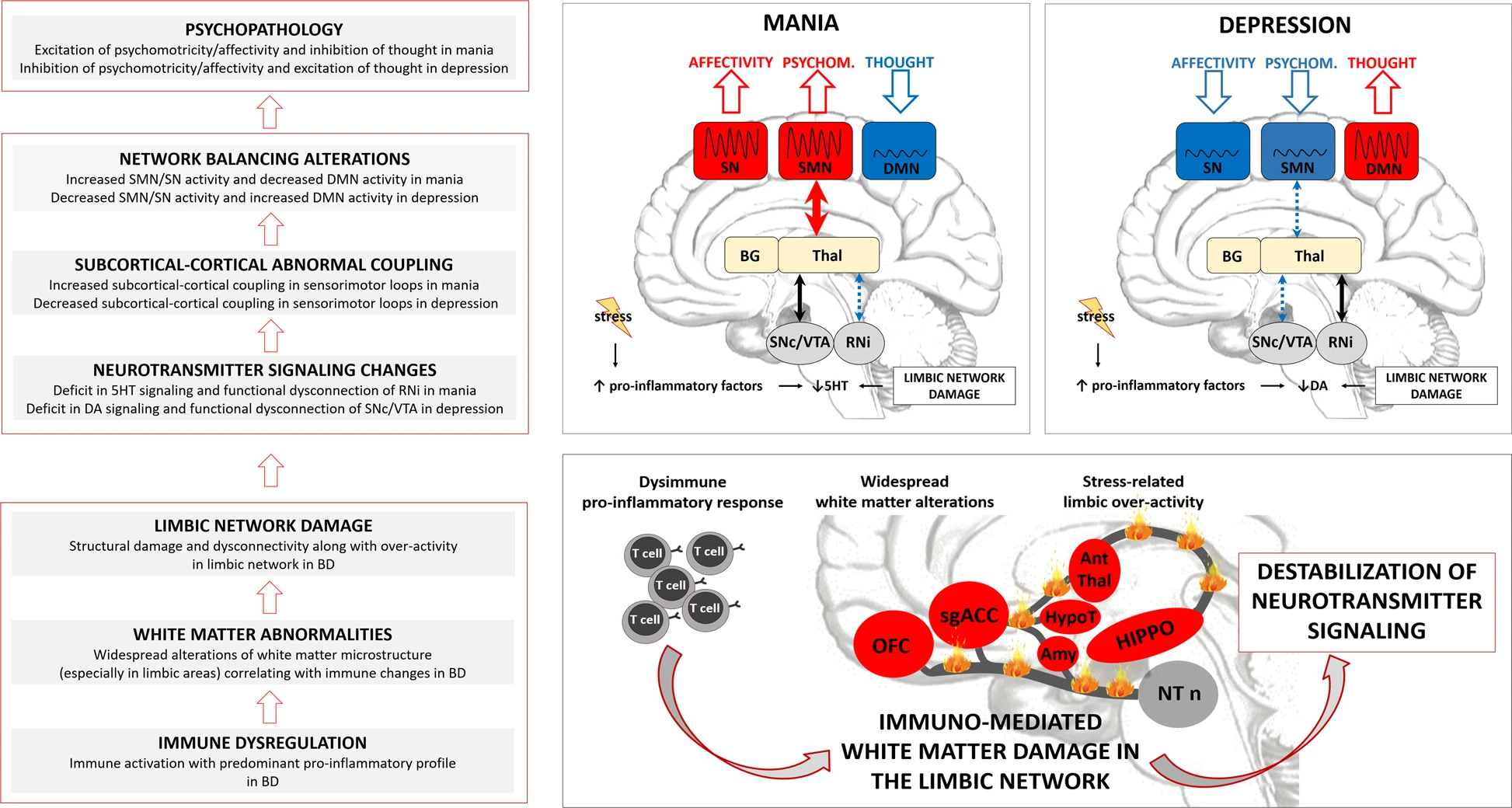
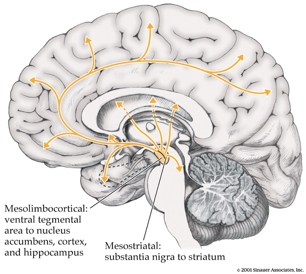
Magioncalda, P., & Martino, M. (2022). A unified model of the pathophysiology of bipolar disorder.
Molecular Psychiatry,
27(1), 202–211.
https://doi.org/10.1038/s41380-021-01091-4
Maletic, V., & Raison, C. (2014). Integrated neurobiology of bipolar disorder.
Frontiers in Psychiatry / Frontiers Research Foundation,
5, 98.
https://doi.org/10.3389/fpsyt.2014.00098
Malhi, G. S., Tanious, M., Das, P., Coulston, C. M., & Berk, M. (2013). Potential
Mechanisms of
Action of
Lithium in
Bipolar Disorder.
CNS Drugs,
27(2), 135–153.
https://doi.org/10.1007/s40263-013-0039-0
MariahCareyVEVO. (2010).
Mariah carey - I’ll be there (MTV unplugged - HD video). You
Tube. Retrieved from
https://www.youtube.com/watch?v=UIt3dx4an9c&list=RDUIt3dx4an9c&start_radio=1
Merikangas, K. R., Akiskal, H. S., Angst, J., Greenberg, P. E., Hirschfeld, R. M. A., Petukhova, M., & Kessler, R. C. (2007). Lifetime and 12-month prevalence of bipolar spectrum disorder in the national comorbidity survey replication.
Archives of General Psychiatry,
64(5), 543–552.
https://doi.org/10.1001/archpsyc.64.5.543
Neuroslicer. (2007).
Schizophrenia: Gerald, part 1. You
Tube. Retrieved from
https://www.youtube.com/watch?v=gGnl8dqEoPQ
Os, J. van, & Kapur, S. (2009). Schizophrenia.
The Lancet,
374(9690), 635–645.
https://doi.org/10.1016/S0140-6736(09)60995-8
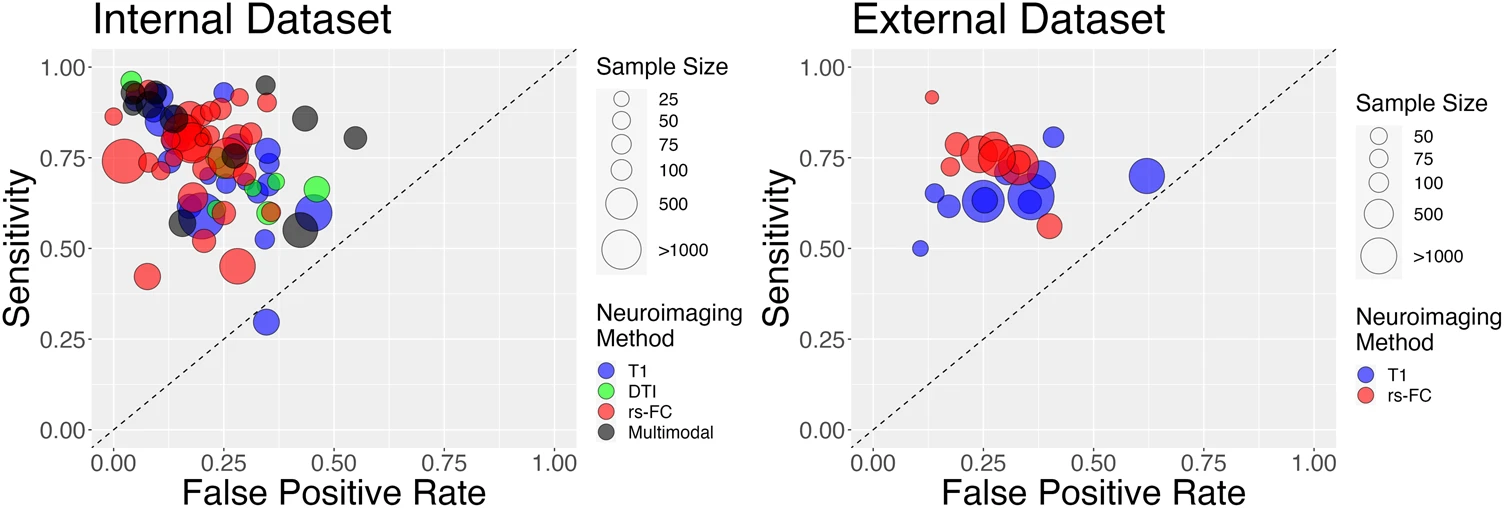
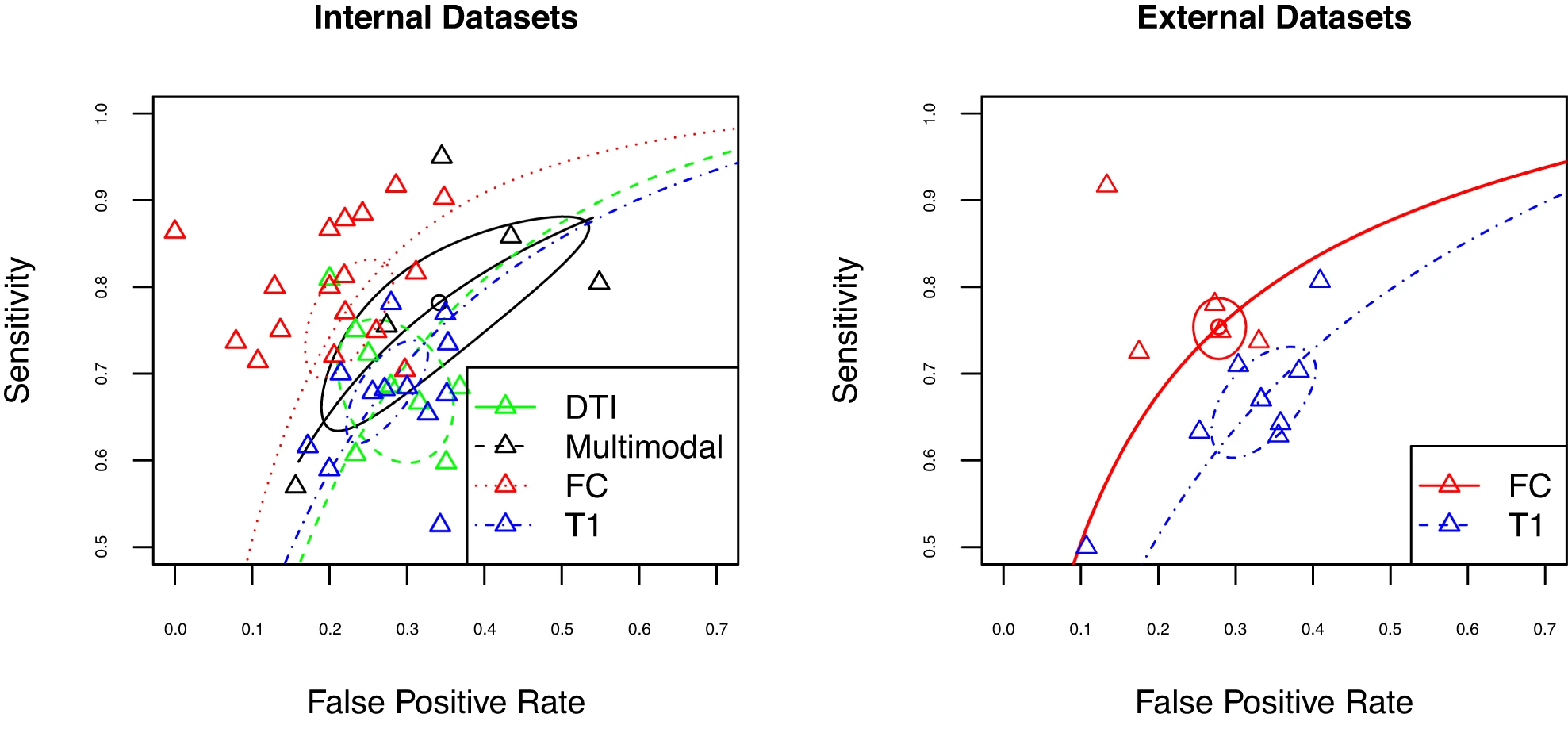
Porter, A., Fei, S., Damme, K. S. F., Nusslock, R., Gratton, C., & Mittal, V. A. (2023). A meta-analysis and systematic review of single vs. Multimodal neuroimaging techniques in the classification of psychosis.
Molecular Psychiatry,
28(8), 3278–3292.
https://doi.org/10.1038/s41380-023-02195-9
Seutin, V. (2005). Dopaminergic neurones: Much more than dopamine?
Br. J. Pharmacol.,
146(2), 167–169.
https://doi.org/10.1038/sj.bjp.0706328
Sidor, M. M., & MacQueen, G. M. (2012). An update on antidepressant use in bipolar depression.
Current Psychiatry Reports,
14(6), 696–704.
https://doi.org/10.1007/s11920-012-0323-6
Sleigh, J., Harvey, M., Voss, L., & Denny, B. (2014). Ketamine – more mechanisms of action than just
NMDA blockade.
Trends in Anaesthesia and Critical Care,
4, 76–81.
https://doi.org/10.1016/j.tacc.2014.03.002
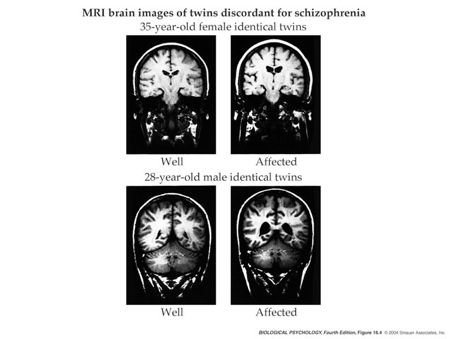
Suddath, R. L., Christison, G. W., Torrey, E. F., Casanova, M. F., & Weinberger, D. R. (1990). Anatomical abnormalities in the brains of monozygotic twins discordant for schizophrenia.
The New England Journal of Medicine,
322(12), 789–794.
https://doi.org/10.1056/NEJM199003223221201
Supekar, K., Los Angeles, C. de, Ryali, S., Kushan, L., Schleifer, C., Repetto, G., … Menon, V. (2024). Robust and replicable functional brain signatures of 22q11.2 deletion syndrome and associated psychosis:
A deep neural network-based multi-cohort study.
Molecular Psychiatry.
https://doi.org/10.1038/s41380-024-02495-8
Tamminga, C. A., Pearlson, G., Keshavan, M., Sweeney, J., Clementz, B., & Thaker, G. (2014). Bipolar and schizophrenia network for intermediate phenotypes: Outcomes across the psychosis continuum.
Schizophrenia Bulletin,
40 Suppl 2(Suppl 2), S131–7.
https://doi.org/10.1093/schbul/sbt179
TheMentallight. (2010).
Schizophrenia ABC 20-20 documentary part 2. You
Tube. Retrieved from
https://www.youtube.com/watch?v=YXimT5CHCDE
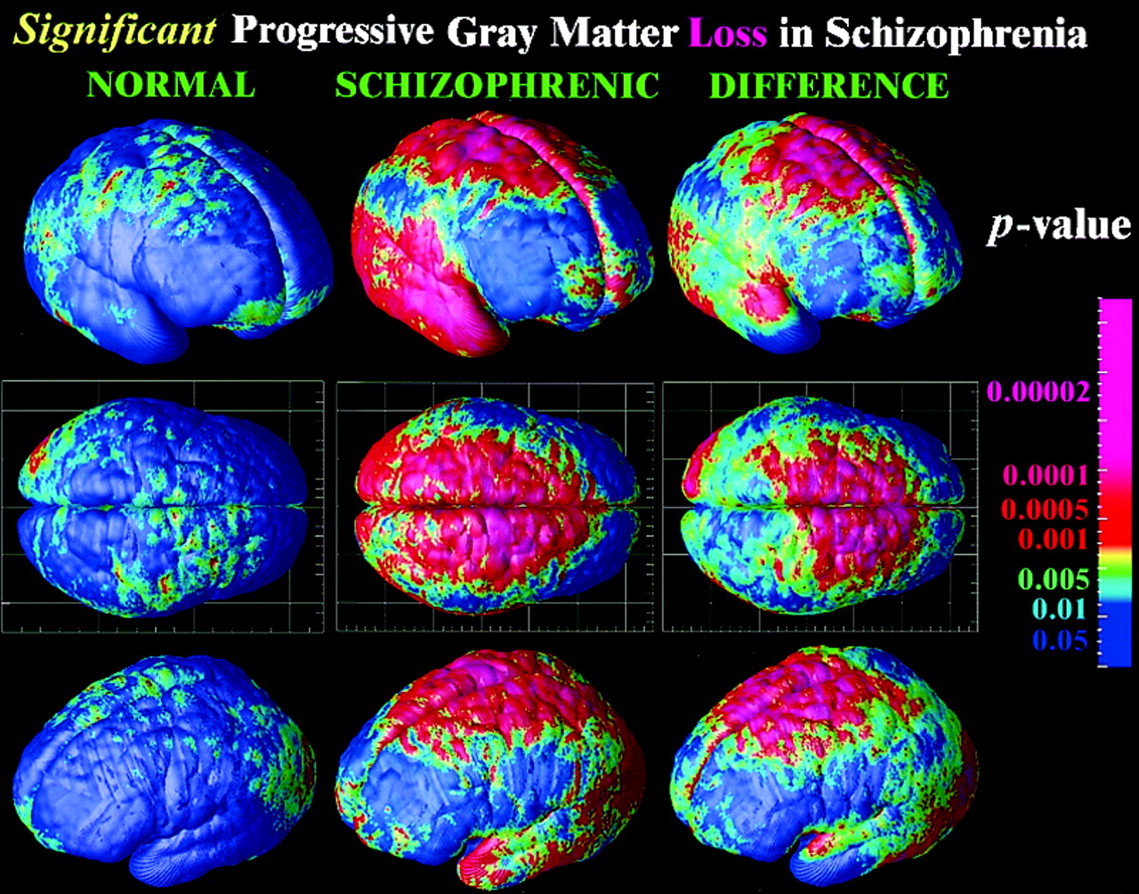
Thompson, P. M., Vidal, C., Giedd, J. N., Gochman, P., Blumenthal, J., Nicolson, R., … Rapoport, J. L. (2001). Mapping adolescent brain change reveals dynamic wave of accelerated gray matter loss in very early-onset schizophrenia.
Proceedings of the National Academy of Sciences,
98(20), 11650–11655.
https://doi.org/10.1073/pnas.201243998
Uhlhaas, P. J. (2013). Dysconnectivity, large-scale networks and neuronal dynamics in schizophrenia.
Curr. Opin. Neurobiol.,
23(2), 283–290.
https://doi.org/10.1016/j.conb.2012.11.004
Vassos, E., Pedersen, C. B., Murray, R. M., Collier, D. A., & Lewis, C. M. (2012). Meta-analysis of the association of urbanicity with schizophrenia.
Schizophrenia Bulletin,
38, 1118–1123.
https://doi.org/10.1093/schbul/sbs096
Yang, G. J., Murray, J. D., Repovs, G., Cole, M. W., Savic, A., Glasser, M. F., … Anticevic, A. (2014). Altered global brain signal in schizophrenia.
Proc. Natl. Acad. Sci. U. S. A.,
111(20), 7438–7443.
https://doi.org/10.1073/pnas.1405289111
Zhang, Y., Ye, F., Zhang, T., Lv, S., Zhou, L., Du, D., … Zhu, S. (2021). Structural basis of ketamine action on human
NMDA receptors.
Nature,
596, 301–305.
https://doi.org/10.1038/s41586-021-03769-9