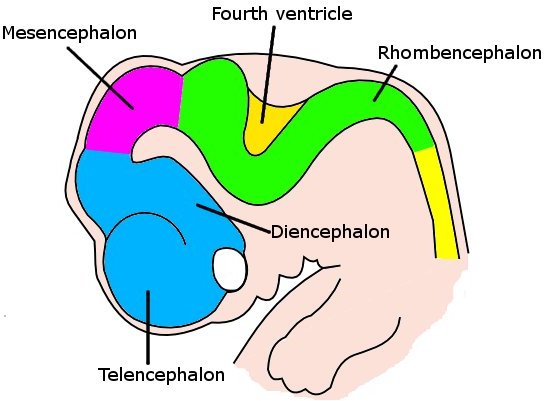
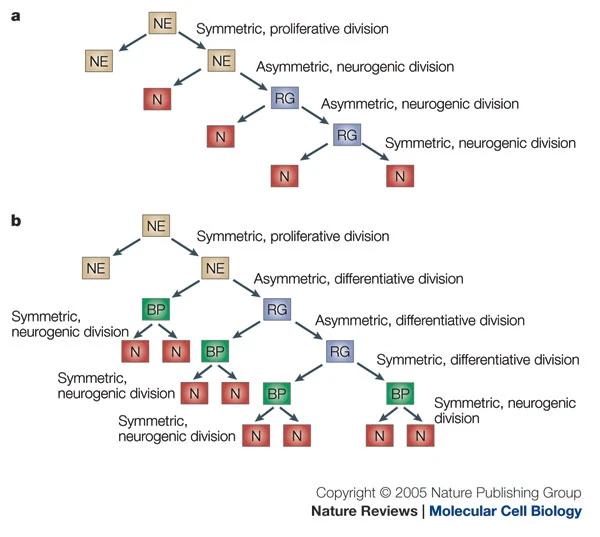
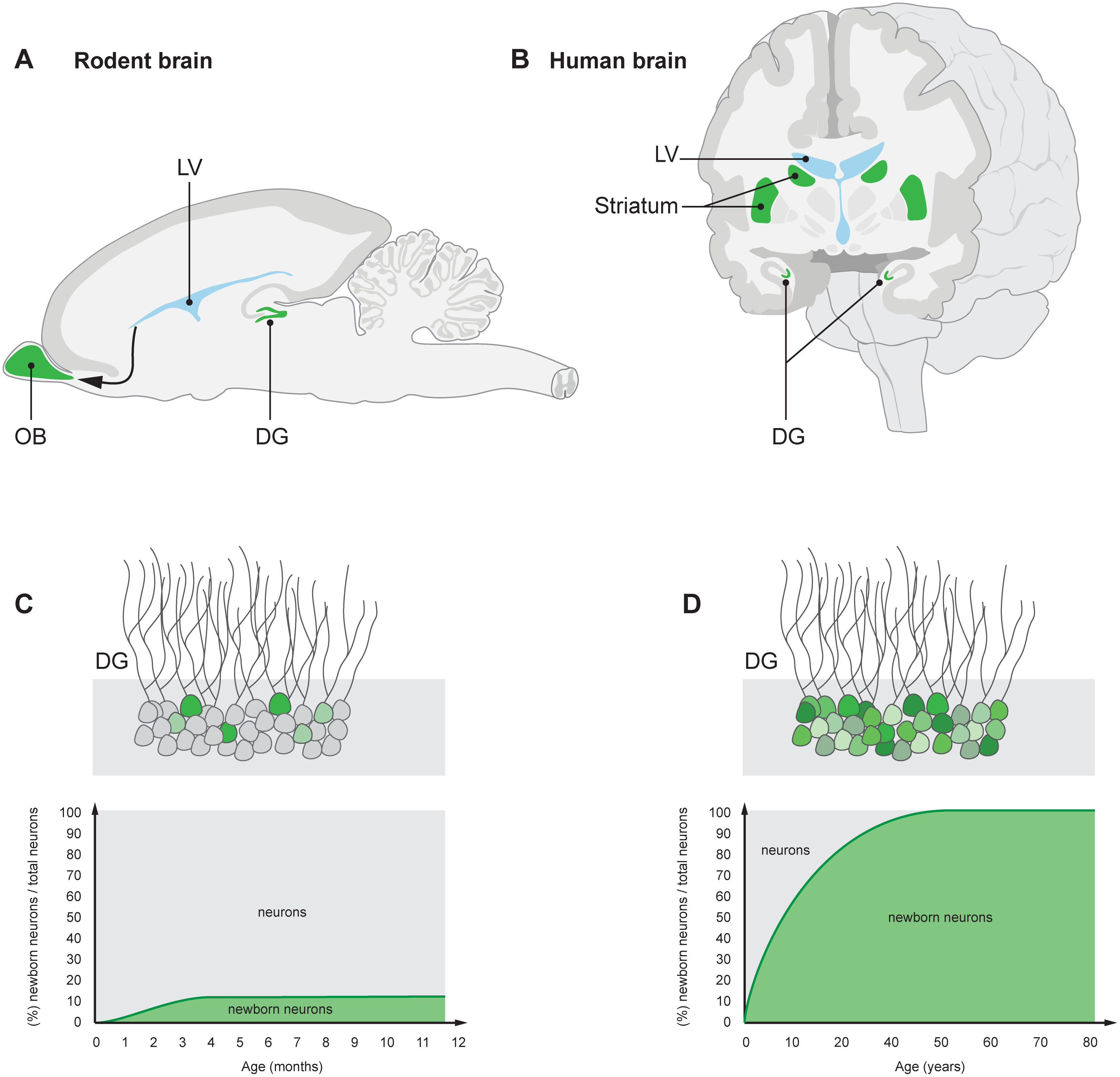
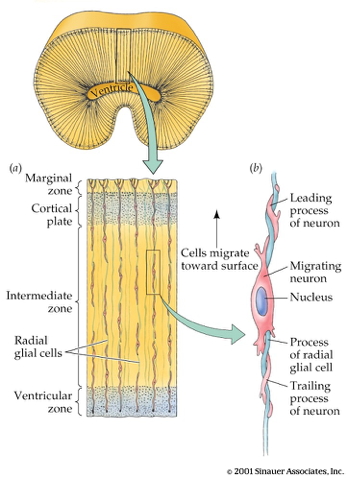
acapellascience. (2017, September).
Evo-Devo (despacito biology parody) | a capella science. Youtube. Retrieved from
https://www.youtube.com/watch?v=ydqReeTV_vk
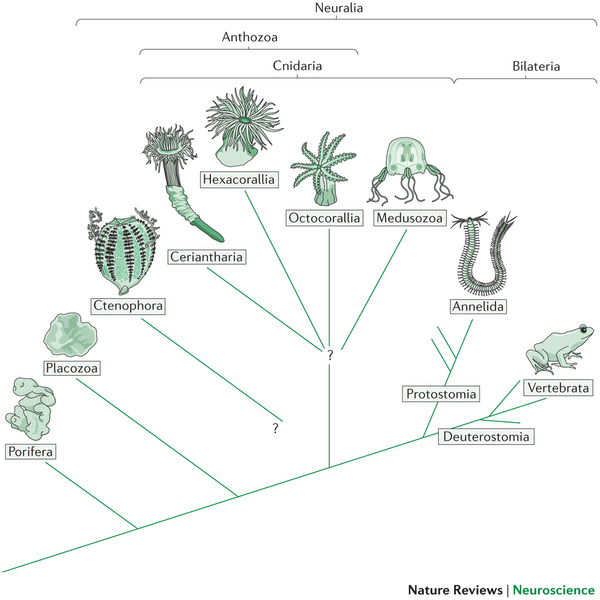
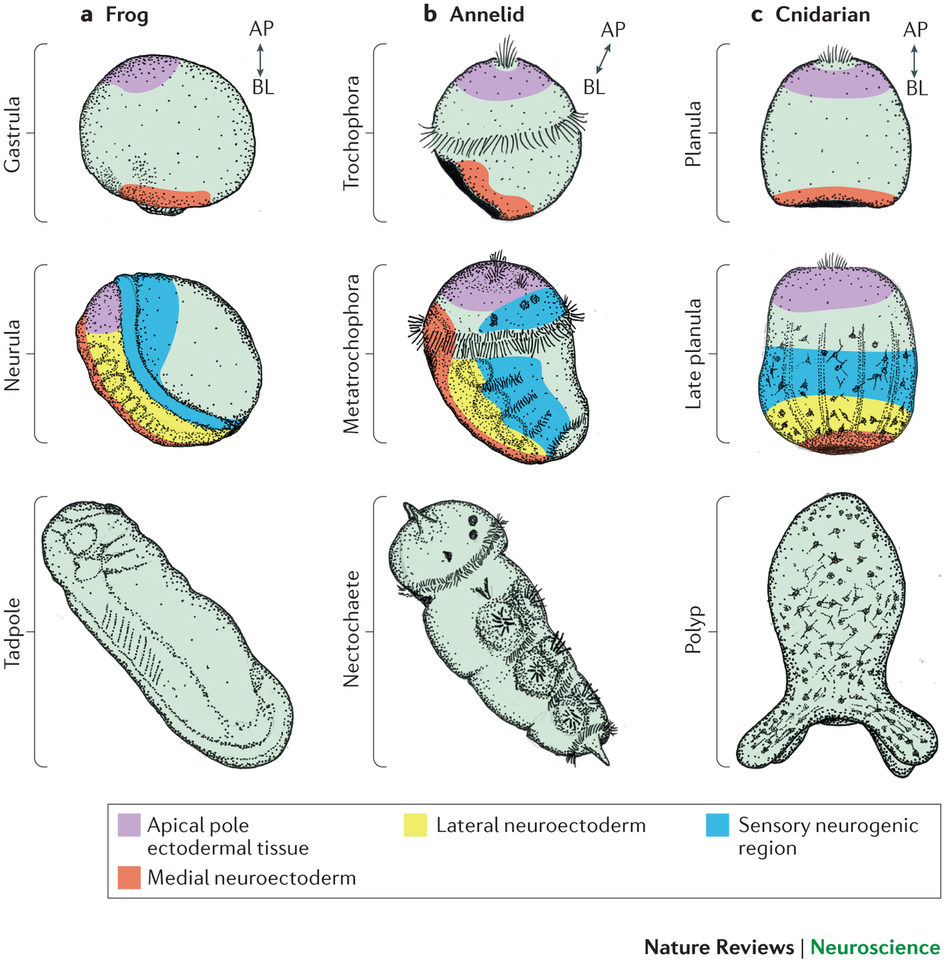
Arendt, D., Tosches, M. A., & Marlow, H. (2016). From nerve net to nerve ring, nerve cord and brain — evolution of the nervous system.
Nature Reviews Neuroscience,
17(1), 61–72.
https://doi.org/10.1038/nrn.2015.15
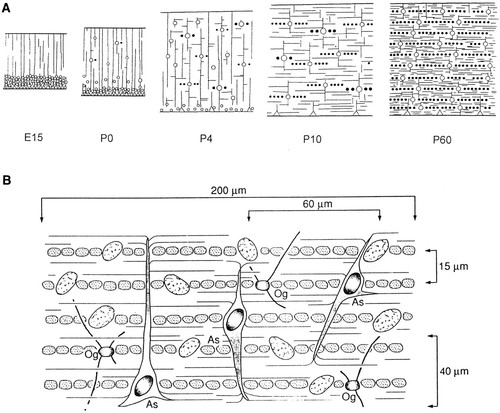
Baumann, N., & Pham-Dinh, D. (2001). Biology of oligodendrocyte and myelin in the mammalian central nervous system.
Physiological Reviews,
81(2), 871–927.
https://doi.org/10.1152/physrev.2001.81.2.871
Blumberg, M. S., & Adolph, K. E. (2023a). Infant action and cognition:
What’s at stake?
Trends in Cognitive Sciences,
27(8), 696–698.
https://doi.org/10.1016/j.tics.2023.05.008
Blumberg, M. S., & Adolph, K. E. (2023b). Protracted development of motor cortex constrains rich interpretations of infant cognition.
Trends in Cognitive Sciences,
27(3), 233–245.
https://doi.org/10.1016/j.tics.2022.12.014
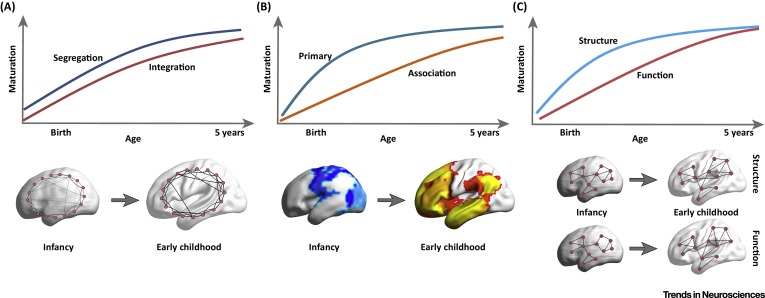
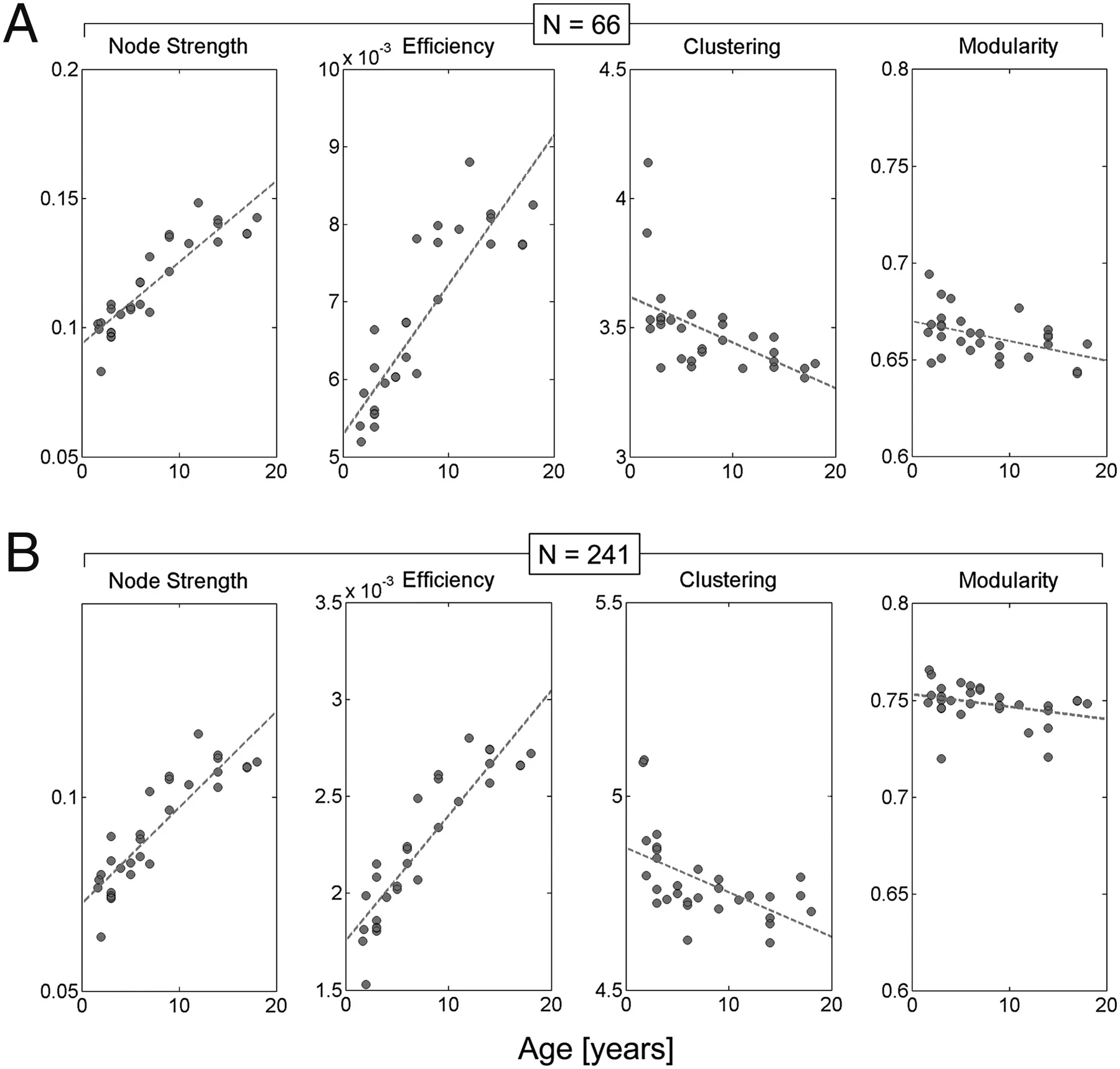
Cao, M., Huang, H., & He, Y. (2017). Developmental connectomics from infancy through early childhood.
Trends in Neuroscience,
40(8), 494–506.
https://doi.org/10.1016/j.tins.2017.06.003
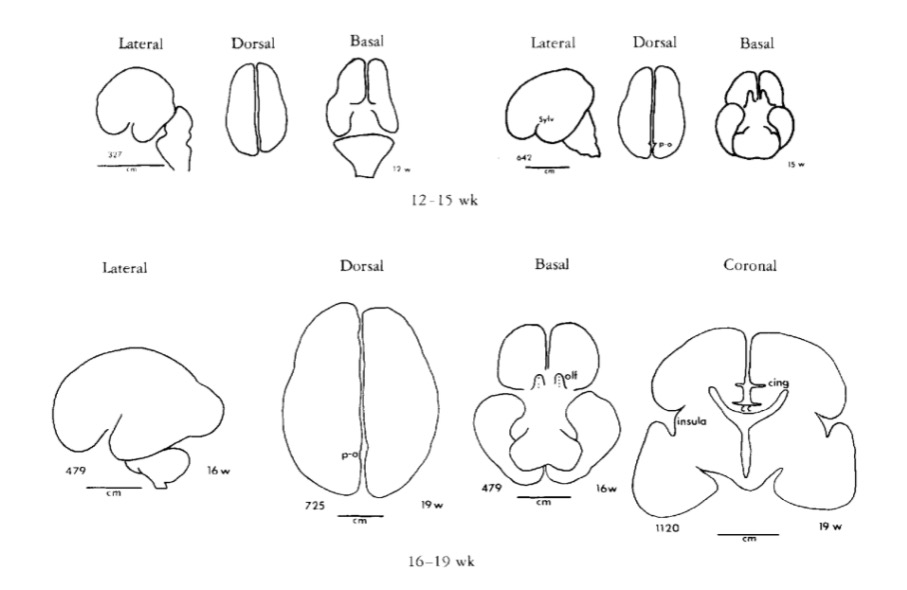
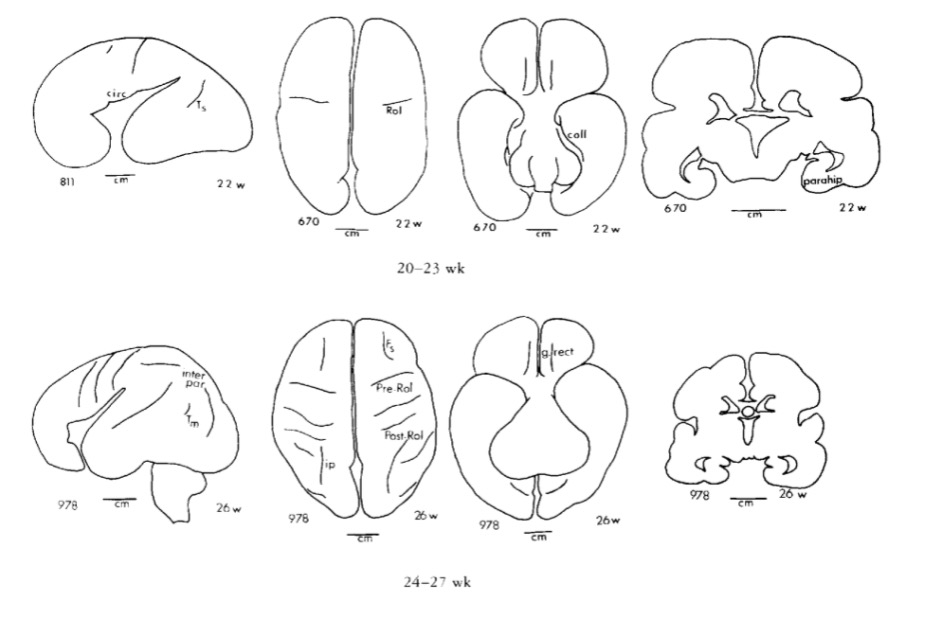
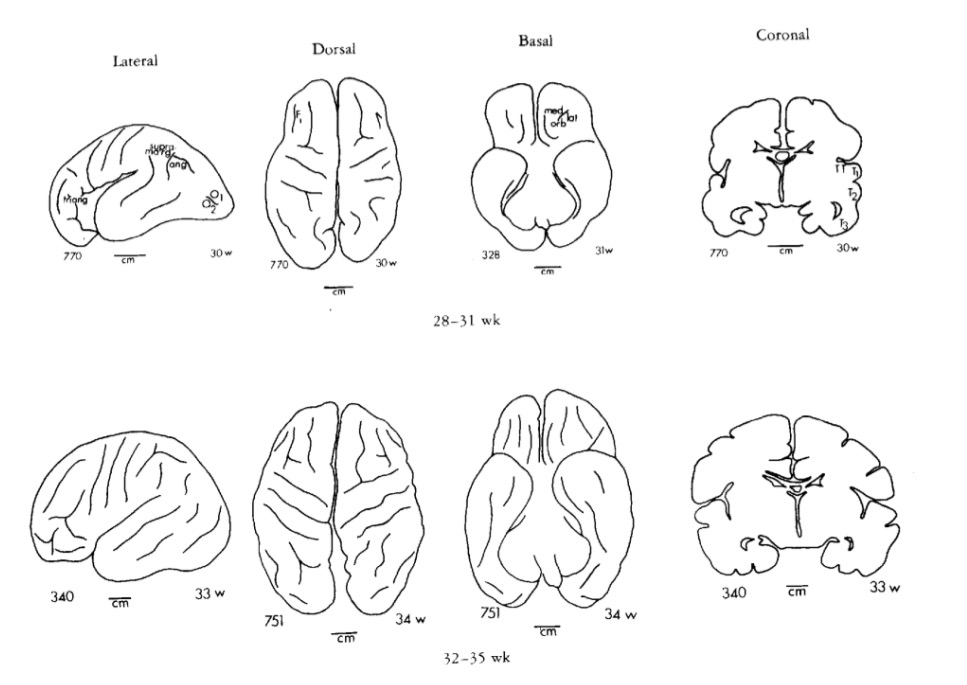
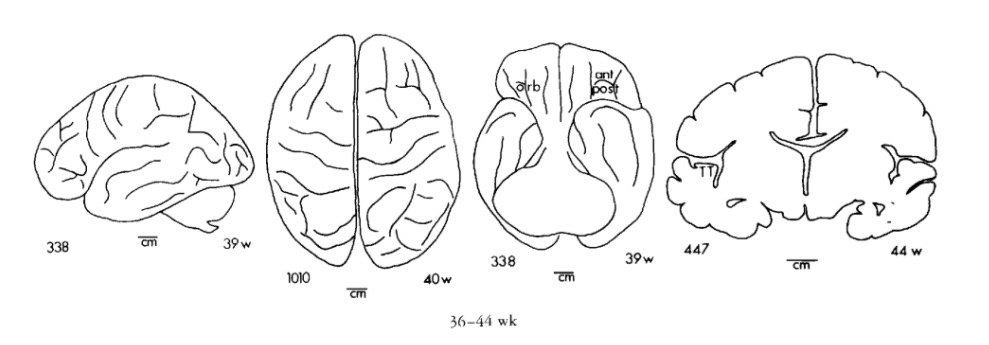
Chi, J. G., Dooling, E. C., & Gilles, F. H. (1977). Gyral development of the human brain.
Ann. Neurol.,
1(1), 86–93.
https://doi.org/10.1002/ana.410010109
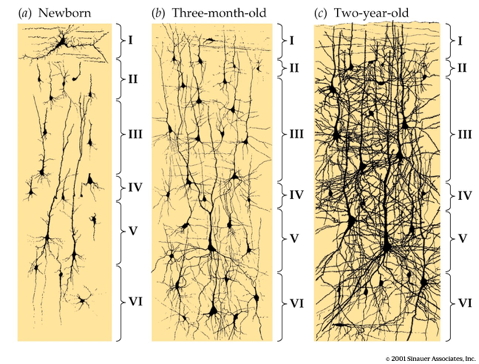
DeFelipe, J., Alonso-Nanclares, L., & Arellano, J. I. (2002). Microstructure of the neocortex: Comparative aspects.
Journal of Neurocytology,
31(3-5), 299–316.
https://doi.org/10.1023/a:1024130211265
Disouky, A., Sanborn, M. A., Sabitha, K. R., Mostafa, M. M., Ayala, I. A., Bennett, D. A., … Lazarov, O. (2026). Human hippocampal neurogenesis in adulthood, ageing and alzheimer’s disease.
Nature, 1–10.
https://doi.org/10.1038/s41586-026-10169-4
Dobzhansky, T. (1973). Nothing in biology makes sense except in the light of evolution.
The American Biology Teacher,
35(3), pp. 125–129. Retrieved from
http://www.jstor.org/stable/4444260
Dunbar, R. I. M. (1998). The social brain hypothesis.
Evolutionary Anthropology,
6, 178–190.
https://doi.org/10.1002/(sici)1520-6505(1998)6:5<178::aid-evan5>3.0.co;2-8
Ernst, A., & Frisén, J. (2015). Adult neurogenesis in humans- common and unique traits in mammals.
PLoS Biology,
13(1), e1002045.
https://doi.org/10.1371/journal.pbio.1002045
Gogtay, N., Giedd, J. N., Lusk, L., Hayashi, K. M., Greenstein, D., Vaituzis, A. C., … Thompson, P. M. (2004). Dynamic mapping of human cortical development during childhood through early adulthood.
Proc. Natl. Acad. Sci. U. S. A.,
101(21), 8174–8179.
https://doi.org/10.1073/pnas.0402680101
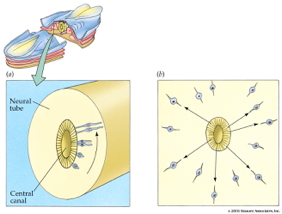
Götz, M., & Huttner, W. B. (2005). The cell biology of neurogenesis.
Nat. Rev. Mol. Cell Biol.,
6(10), 777–788.
https://doi.org/10.1038/nrm1739
Haeckel, W. by E. (2001, August 12). Ernst
Haeckel. Retrieved March 18, 2026, from
https://en.wikipedia.org/wiki/Ernst_Haeckel
Hagmann, P., Sporns, O., Madan, N., Cammoun, L., Pienaar, R., Wedeen, V. J., … Grant, P. E. (2010). White matter maturation reshapes structural connectivity in the late developing human brain.
Proceedings of the National Academy of Sciences,
107(44), 19067–19072.
https://doi.org/10.1073/pnas.1009073107
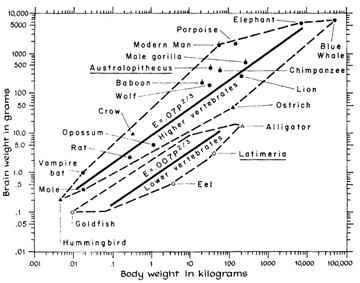
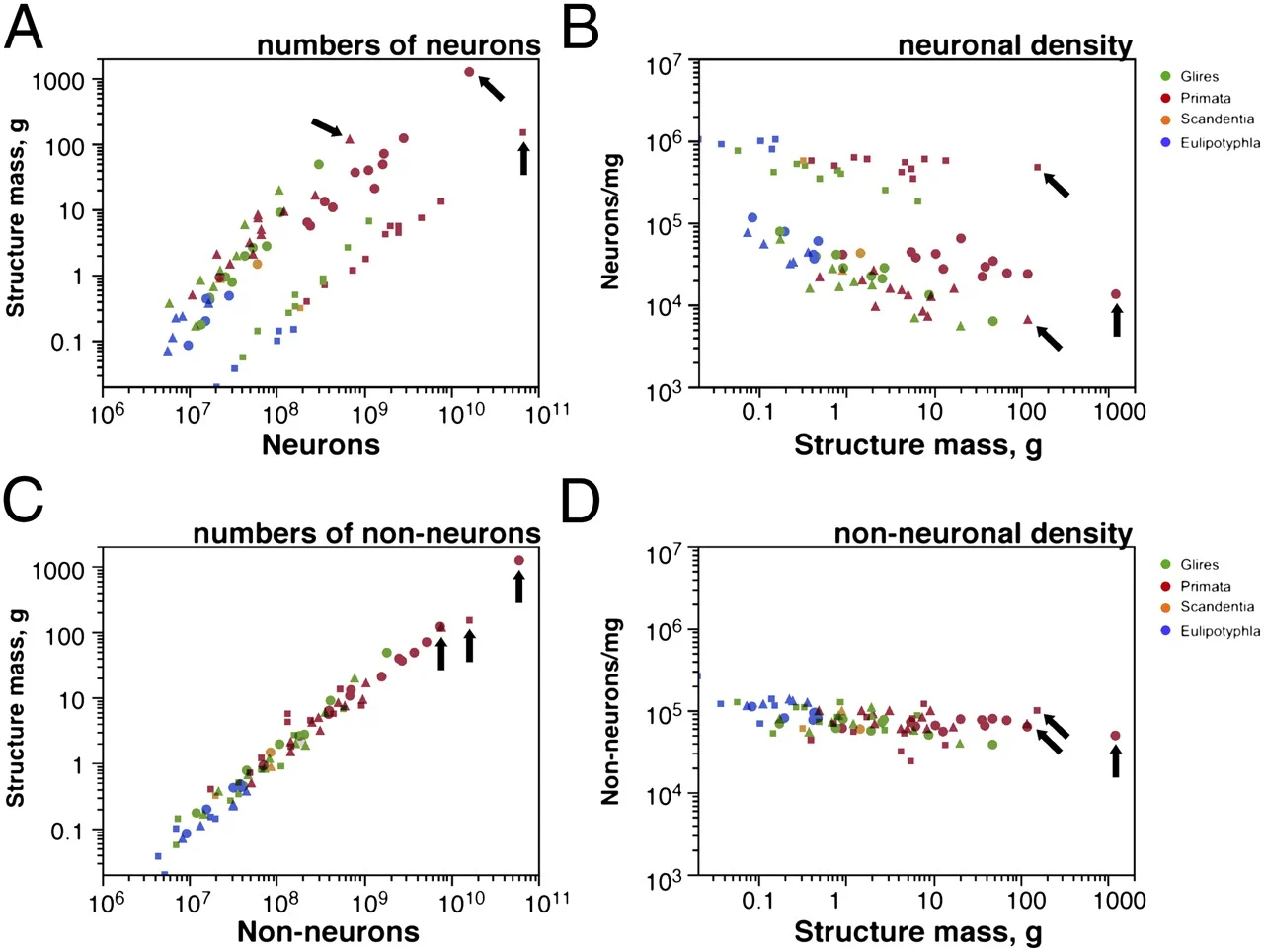
Herculano-Houzel, S. (2012). The remarkable, yet not extraordinary, human brain as a scaled-up primate brain and its associated cost.
Proceedings of the National Academy of Sciences of the United States of America,
109 Suppl 1, 10661–10668.
https://doi.org/10.1073/pnas.1201895109
Herculano-Houzel, S. (2016).
The human advantage: A new understanding of how our brain became remarkable. MIT Press. Retrieved from
https://market.android.com/details?id=book-DMqpCwAAQBAJ
Herculano-Houzel, S. (2017). Numbers of neurons as biological correlates of cognitive capability.
Current Opinion in Behavioral Sciences,
16(Supplement C), 1–7.
https://doi.org/10.1016/j.cobeha.2017.02.004
Hofman, M. A. (2014). Evolution of the human brain: When bigger is better.
Frontiers in Neuroanatomy,
8.
https://doi.org/10.3389/fnana.2014.00015
Huttenlocher, P. R. (2009).
Neural plasticity: The effects of environment on the development of the cerebral cortex. Harvard University Press. Retrieved from
https://play.google.com/store/books/details?id=2uhP2CBnaGwC
Irimia, A., & Van Horn, J. (2014). Systematic network lesioning reveals the core white matter scaffold of the human brain.
Frontiers in Human Neuroscience,
8, 51.
https://doi.org/10.3389/fnhum.2014.00051
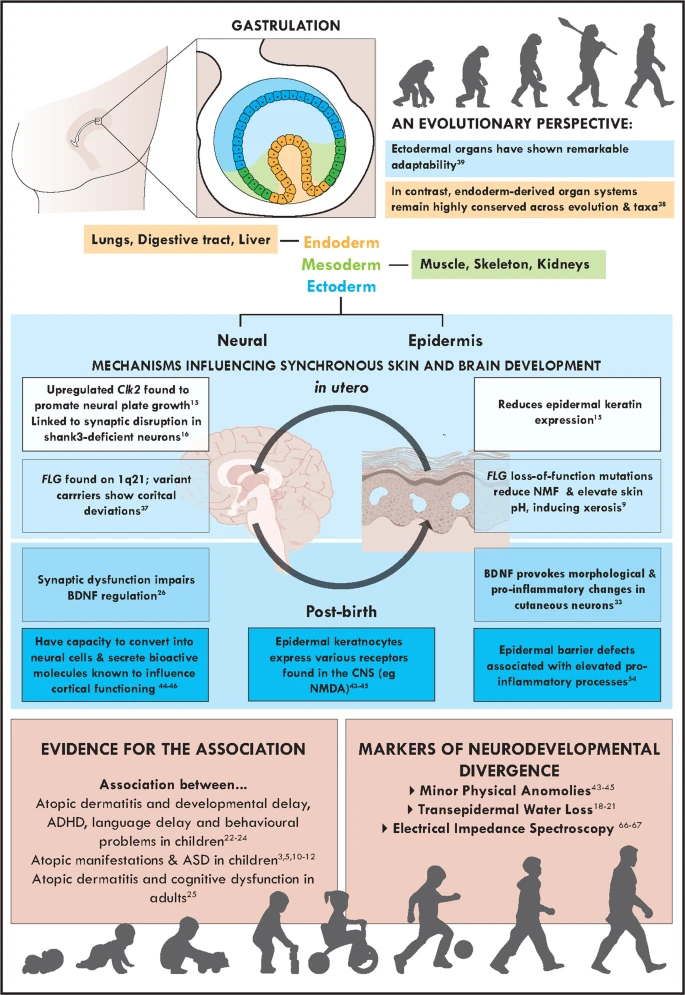
Jameson, C., Boulton, K. A., Silove, N., Nanan, R., & Guastella, A. J. (2023). Ectodermal origins of the skin-brain axis: A novel model for the developing brain, inflammation, and neurodevelopmental conditions.
Molecular Psychiatry,
28(1), 108–117.
https://doi.org/10.1038/s41380-022-01829-8
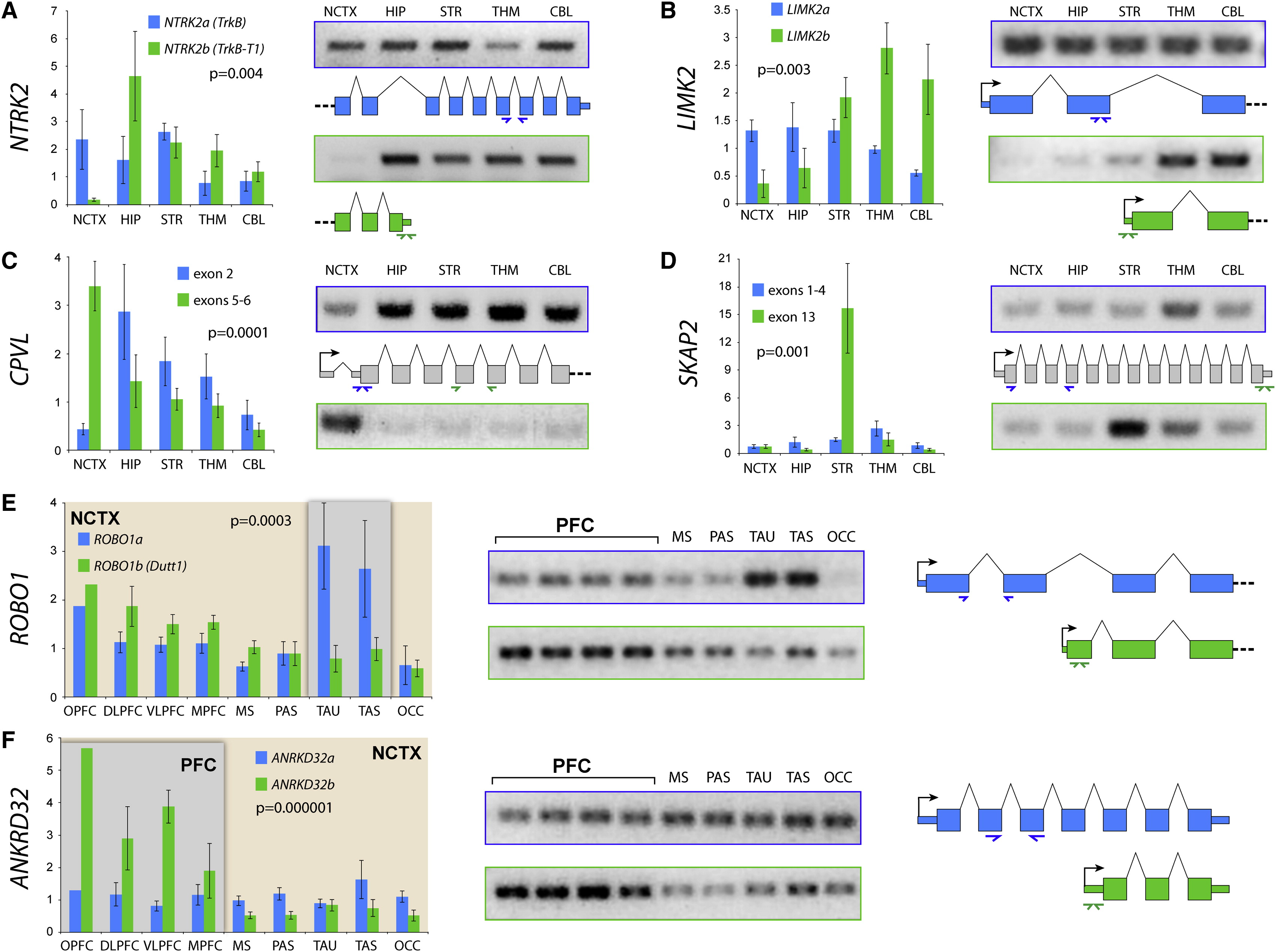
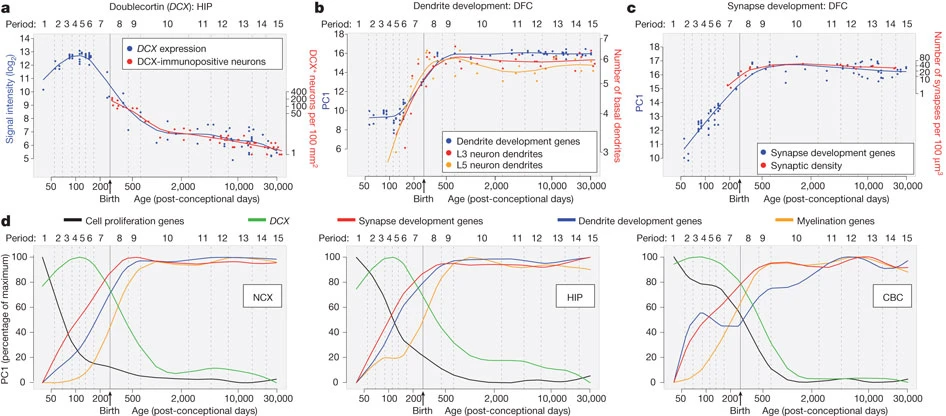
Johnson, M. B., Kawasawa, Y. I., Mason, C. E., Krsnik, Ž., Coppola, G., Bogdanović, D., … Šestan, N. (2009). Functional and evolutionary insights into human brain development through global transcriptome analysis. Neuron, 62(4), 494–509.
Johnson, M. H. (2001). Functional brain development in humans.
Nat. Rev. Neurosci.,
2(7), 475–483.
https://doi.org/10.1038/35081509
Kang, H. J., Kawasawa, Y. I., Cheng, F., Zhu, Y., Xu, X., Li, M., … Šestan, N. (2011). Spatio-temporal transcriptome of the human brain.
Nature,
478(7370), 483–489.
https://doi.org/10.1038/nature10523
Kety, S. S., & Schmidt, C. F. (1948).
The
Nitrous
OXIDE METHOD FOR THE QUANTITATIVE DETERMINATION OF CEREBRAL BLOOD FLOW IN MAN:
THEORY,
PROCEDURE AND NORMAL VALUES.
The Journal of Clinical Investigation,
27(4), 476–483.
https://doi.org/10.1172/JCI101994
Knickmeyer, R. C., Gouttard, S., Kang, C., Evans, D., Wilber, K., Smith, J. K., … Gilmore, J. H. (2008). A structural
MRI study of human brain development from birth to 2 years.
J. Neurosci.,
28(47), 12176–12182.
https://doi.org/10.1523/JNEUROSCI.3479-08.2008
Konner, M. (2011).
The Evolution of Childhood. Belknap Press of Harvard University Press. Retrieved from
http://www.hup.harvard.edu/catalog.php?isbn=9780674062016
Kuzawa, C. W., Chugani, H. T., Grossman, L. I., Lipovich, L., Muzik, O., Hof, P. R., … Lange, N. (2014). Metabolic costs and evolutionary implications of human brain development.
Proc. Natl. Acad. Sci. U. S. A.,
111(36), 13010–13015.
https://doi.org/10.1073/pnas.1323099111
Marner, L., Nyengaard, J. R., Tang, Y., & Pakkenberg, B. (2003). Marked loss of myelinated nerve fibers in the human brain with age.
The Journal of Comparative Neurology,
462(2), 144–152.
https://doi.org/10.1002/cne.10714
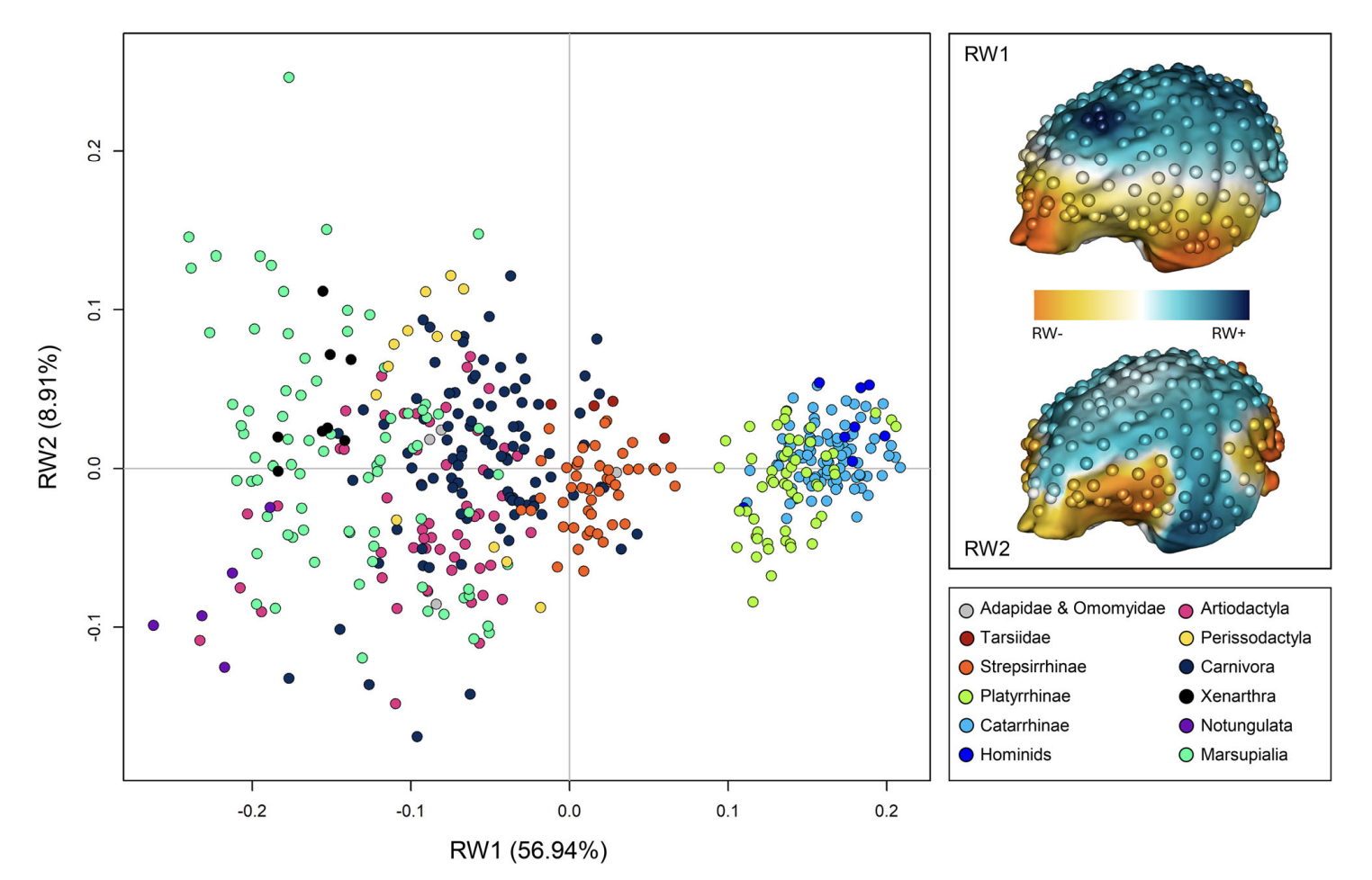
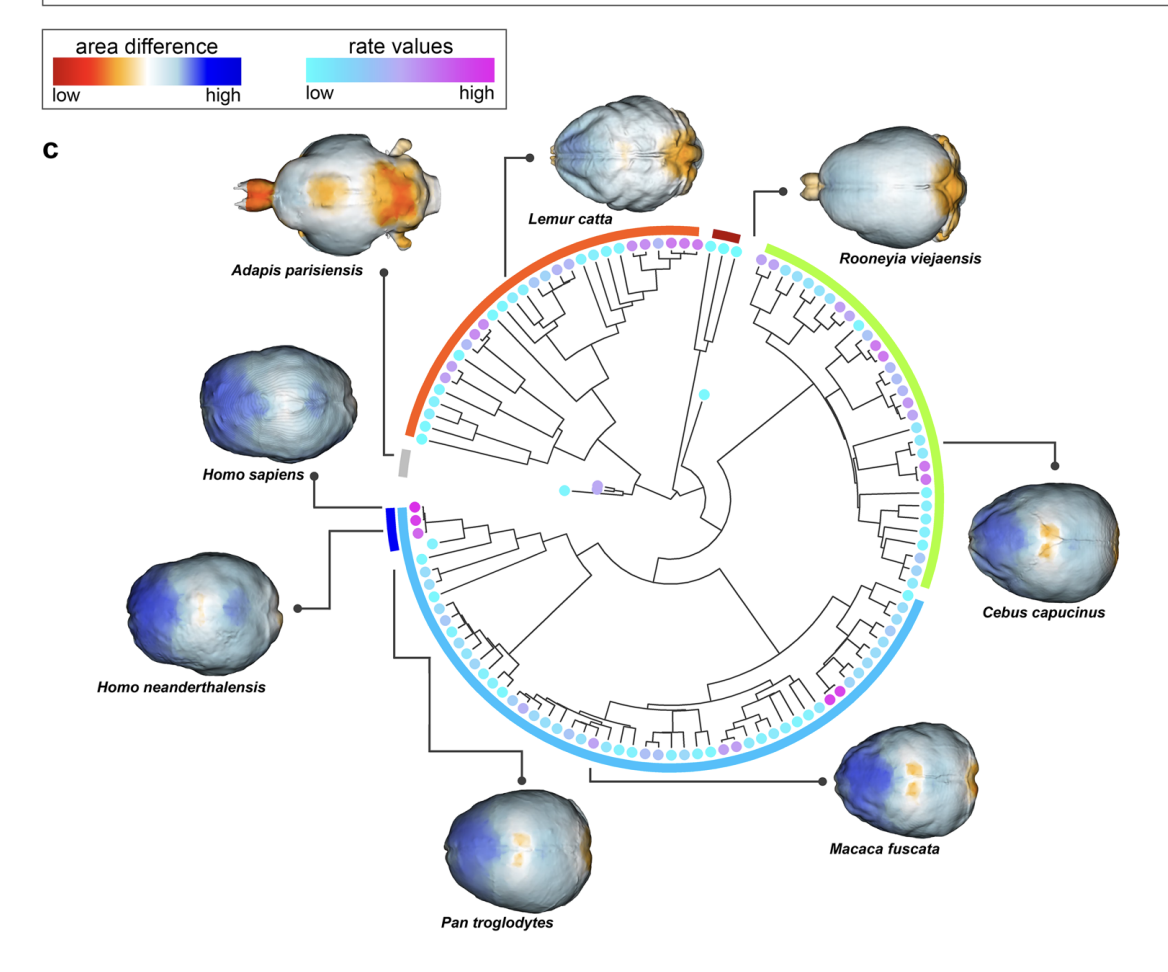
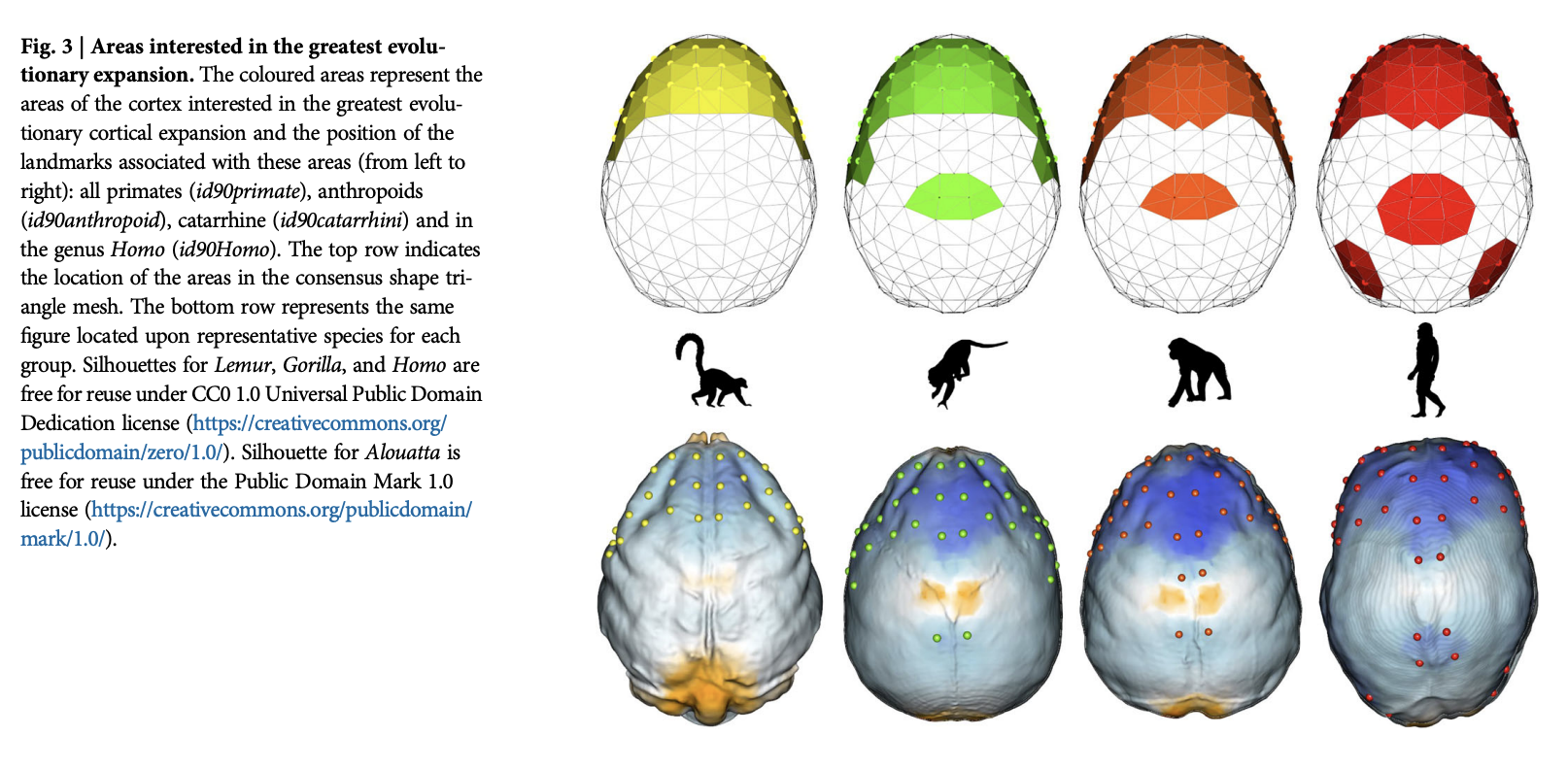
Melchionna, M., Castiglione, S., Girardi, G., Profico, A., Mondanaro, A., Sansalone, G., … Raia, P. (2025). Cortical areas associated to higher cognition drove primate brain evolution.
Communications Biology,
8, 80.
https://doi.org/10.1038/s42003-025-07505-1
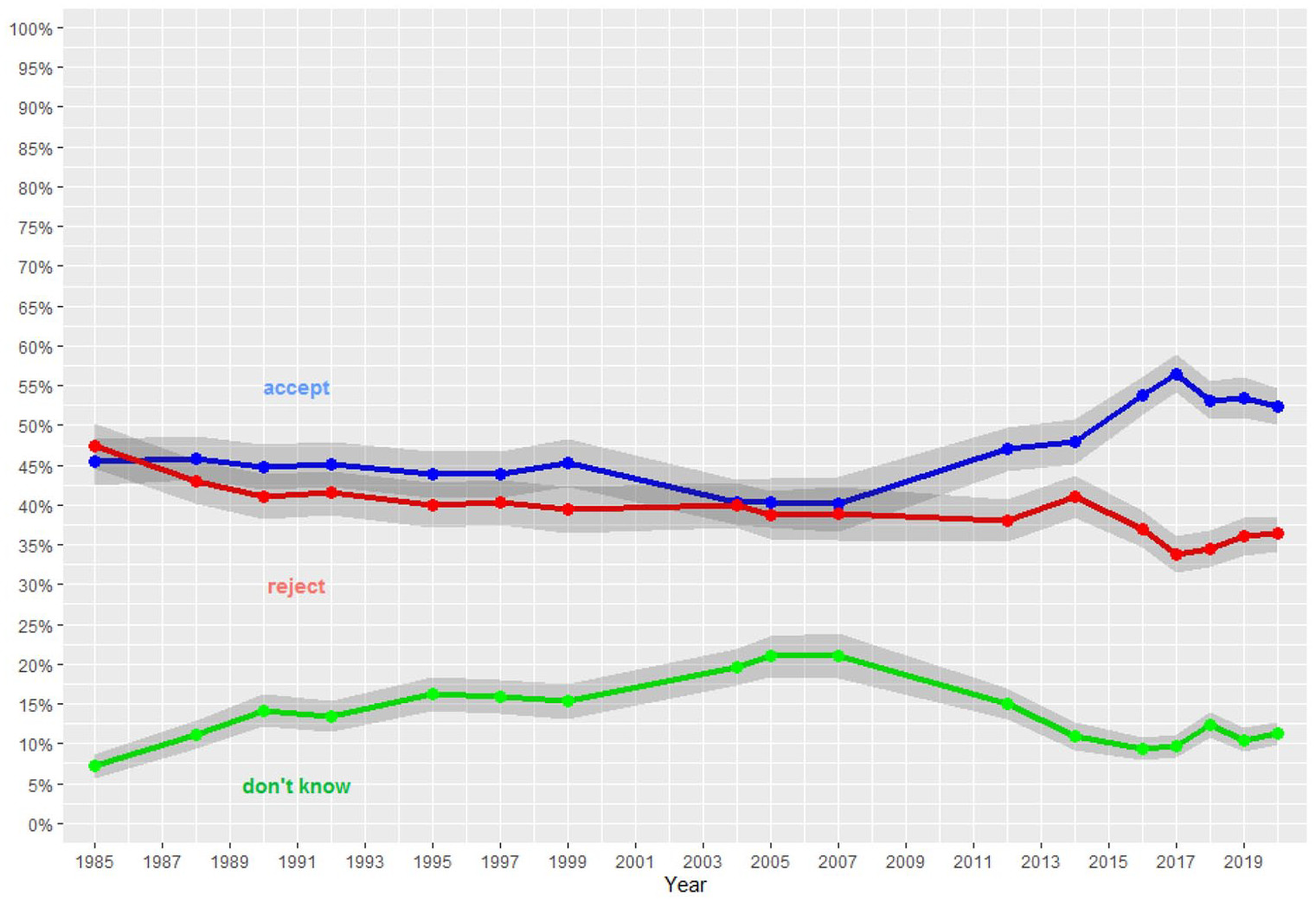
Miller, J. D., Scott, E. C., Ackerman, M. S., Laspra, B., Branch, G., Polino, C., & Huffaker, J. S. (2021). Public acceptance of evolution in the united states, 1985-2020.
Public Understanding of Science, 9636625211035919.
https://doi.org/10.1177/09636625211035919
Miller, J. D., Scott, E. C., & Okamoto, S. (2006). Public acceptance of evolution.
SCIENCE-NEW YORK THEN WASHINGTON-,
313(5788), 765.
https://doi.org/10.1126/science.1126746
Mr Riddz Science. (2015, September). Apoptosis - programmed cell death. Youtube. Retrieved from
https://www.youtube.com/watch?v=ukxNXSPckjQ
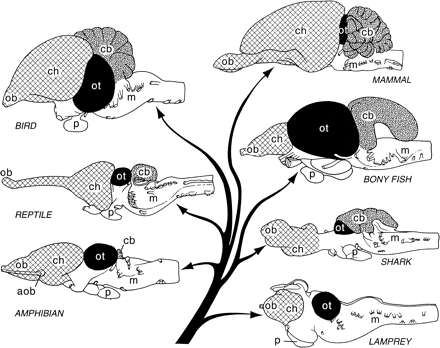
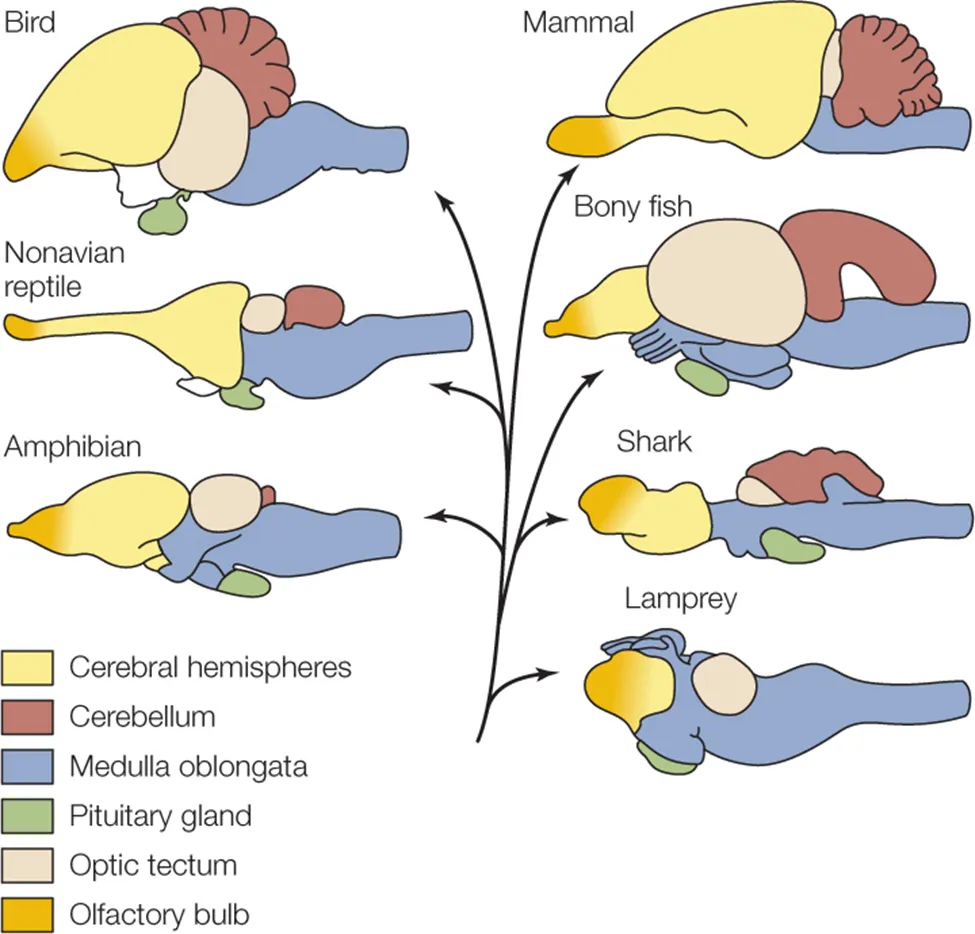
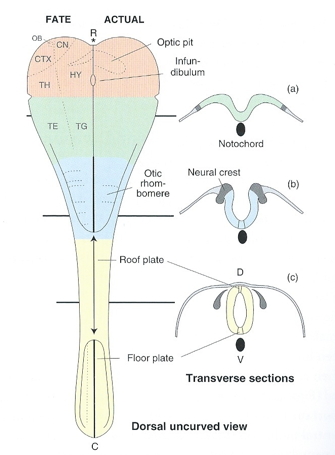
Northcutt, R. G. (2002). Understanding vertebrate brain evolution.
Integr. Comp. Biol.,
42(4), 743–756.
https://doi.org/10.1093/icb/42.4.743
Park, S. (2009).
Hydra movement. You
Tube. Retrieved from
https://www.youtube.com/watch?v=-UI531GMRTM
Petrican, R., Taylor, M. J., & Grady, C. L. (2017). Trajectories of brain system maturation from childhood to older adulthood: Implications for lifespan cognitive functioning.
Neuroimage.
https://doi.org/10.1016/j.neuroimage.2017.09.025
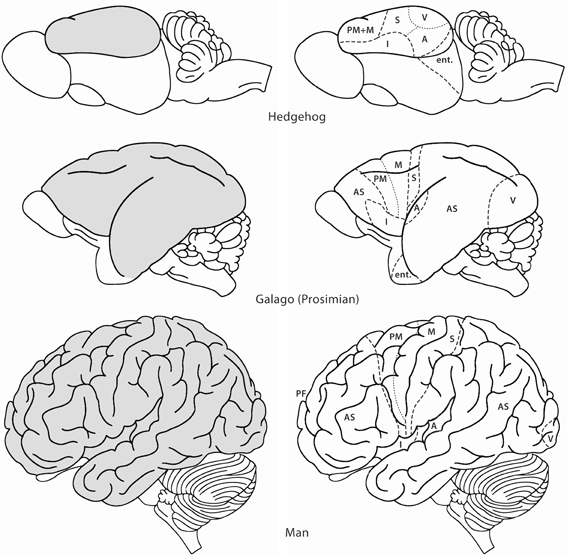
Rakic, P. (2009). Evolution of the neocortex: A perspective from developmental biology.
Nature Reviews. Neuroscience,
10(10), 724–735.
https://doi.org/10.1038/nrn2719
Shaw, P., Kabani, N. J., Lerch, J. P., Eckstrand, K., Lenroot, R., Gogtay, N., … Others. (2008). Neurodevelopmental trajectories of the human cerebral cortex.
Journal of Neuroscience,
28(14), 3586–3594.
https://doi.org/10.1523/JNEUROSCI.5309-07.2008
Silbereis, J. C., Pochareddy, S., Zhu, Y., Li, M., & Sestan, N. (2016). The cellular and molecular landscapes of the developing human central nervous system.
Neuron,
89(2), 248–268.
https://doi.org/10.1016/j.neuron.2015.12.008
Swanson, L. W. (2012). Brain architecture: Understanding the basic plan. Oxford University Press.
twoodburn. (2008).
"Gentle arms of eden", by dave carter and tracy grammer. You
Tube. Retrieved from
https://www.youtube.com/watch?v=bAGMATHlSK4&list=RDbAGMATHlSK4&start_radio=1
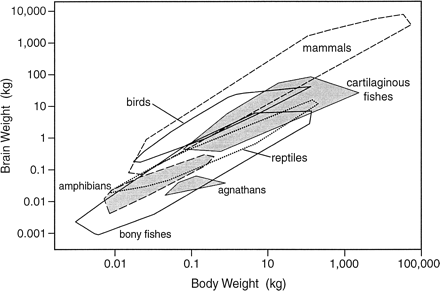
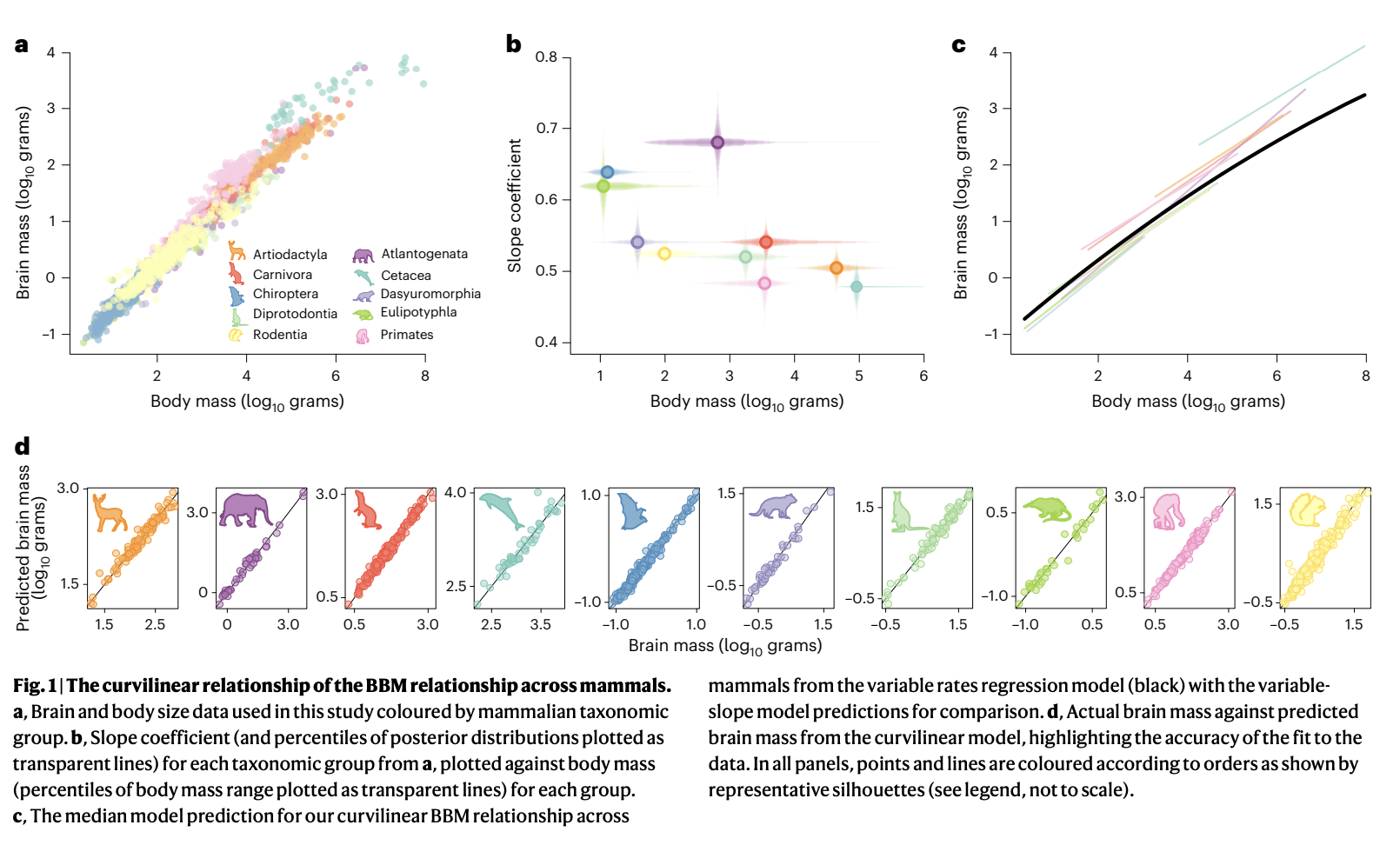
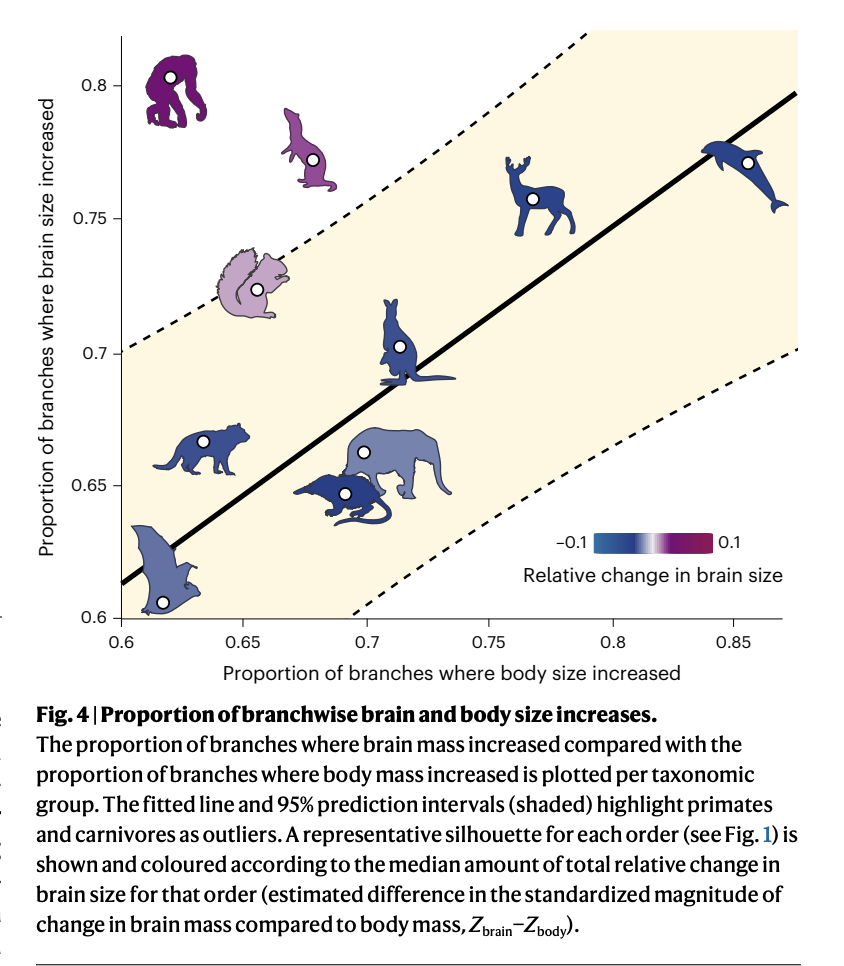
Venditti, C., Baker, J., & Barton, R. A. (2024). Co-evolutionary dynamics of mammalian brain and body size.
Nature Ecology & Evolution,
8, 1534–1542.
https://doi.org/10.1038/s41559-024-02451-3
Wikipedia contributors. (2026, March 14). Nothing in biology makes sense except in the light of evolution. Retrieved from
https://en.wikipedia.org/wiki/Nothing_in_Biology_Makes_Sense_Except_in_the_Light_of_Evolution
Wooster, E. I. F., Whiting, M. J., Nimmo, D. G., Sayol, F., Carthey, A. J. R., Stanton, L. A., & Ashton, B. J. (2026). Predator–prey interactions as drivers of cognitive evolution.
Nature Reviews Biodiversity, 1–9.
https://doi.org/10.1038/s44358-026-00141-5
Wrangham, R. (2009).
Catching fire: How cooking made us human. Basic Books. Retrieved from
https://market.android.com/details?id=book-ebEOupKz-rMC